2
answers
0
watching
3,908
views
28 Sep 2019
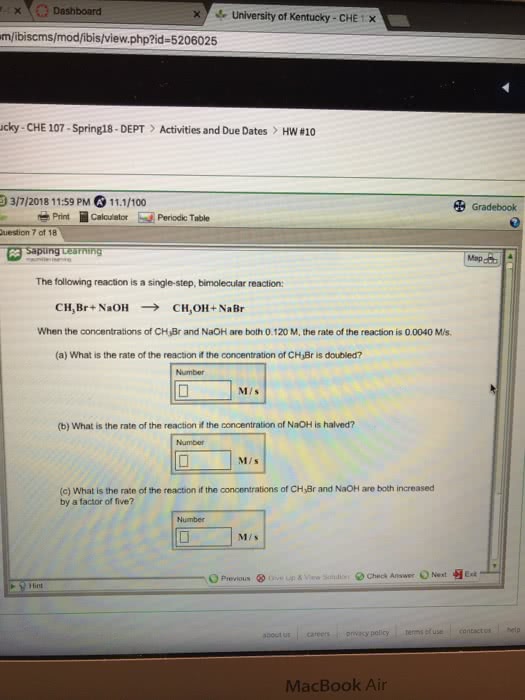
The following reaction is a single-step, bimolecular reaction: CH3Br+NaOH--->CH3OH+NaBr. When the concentrations of CH3Br and NaOH are both 0.170 M, the rate of the reaction is 0.0090 M/s. (a) What is the rate of the reaction if the concentration of CH3Br is doubled? (b) What is the rate of the reaction if the concentration of NaOH is halved? (c) What is the rate of the reaction if the concentrations of CH3Br and NaOH are both increased by a factor of five?
The following reaction is a single-step, bimolecular reaction: CH3Br+NaOH--->CH3OH+NaBr. When the concentrations of CH3Br and NaOH are both 0.170 M, the rate of the reaction is 0.0090 M/s. (a) What is the rate of the reaction if the concentration of CH3Br is doubled? (b) What is the rate of the reaction if the concentration of NaOH is halved? (c) What is the rate of the reaction if the concentrations of CH3Br and NaOH are both increased by a factor of five?
Read by 1 person
15 Feb 2023
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University16 Apr 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.