1
answer
0
watching
130
views
28 Sep 2019
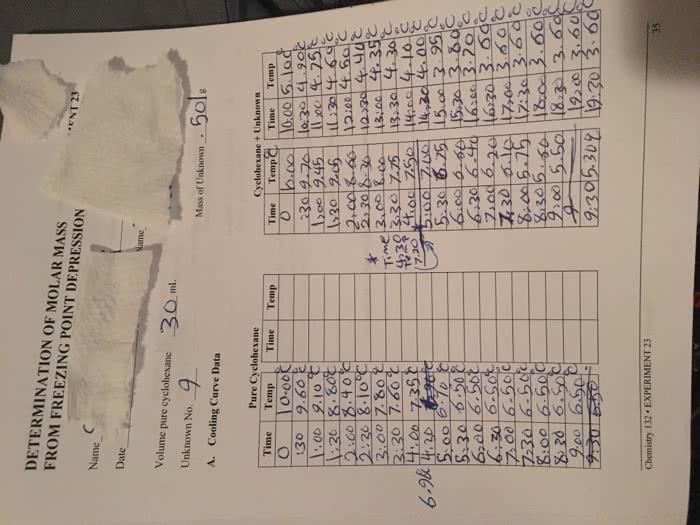
when a solute is added to a solvent, the boiling point of the solution is higher than the pure solvent's was, but the melting point of it is lower. i know this is because the intermolecular forces are larger when it is boiling, and smaller when it is melting. i also know that it has something to do with the different forces between solute-solute, solvent-solvent, and solute-solvent. what i don't understand is why intermolecular forces can be strong at one point, yet weak at others. thanks for the help.
when a solute is added to a solvent, the boiling point of the solution is higher than the pure solvent's was, but the melting point of it is lower. i know this is because the intermolecular forces are larger when it is boiling, and smaller when it is melting. i also know that it has something to do with the different forces between solute-solute, solvent-solvent, and solute-solvent. what i don't understand is why intermolecular forces can be strong at one point, yet weak at others. thanks for the help.
1
answer
0
watching
130
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
28 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232