1
answer
0
watching
1,313
views
28 Sep 2019
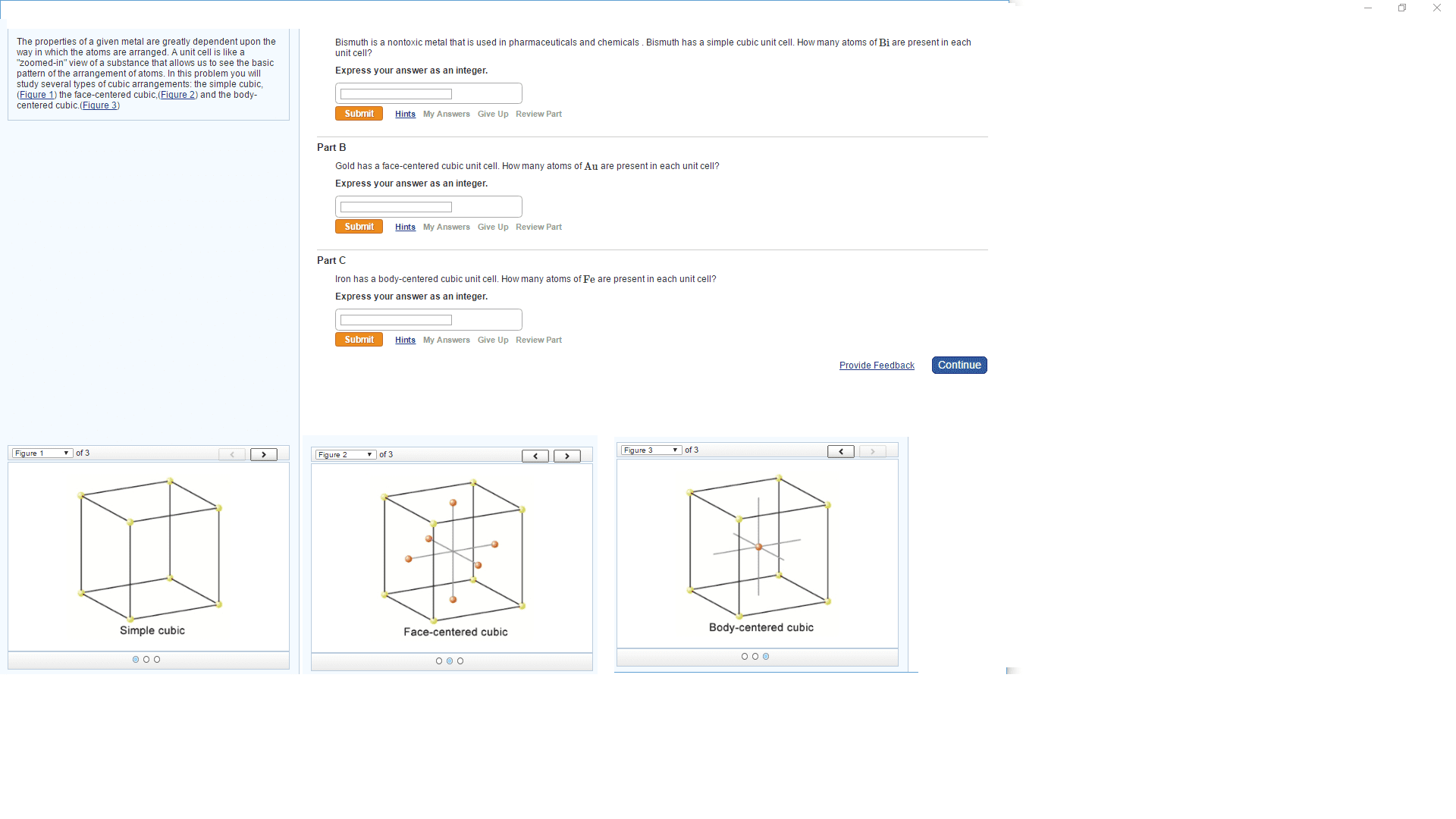
The density of solid Fe is 7.87 g/cm3. How many atoms are presentper cubic centimeter of Fe =__
As a solid Fe adopts a body-centered cubic unit cell. How many unitcells are present per cubic centimeter of Fe,=___What is the volume of a unit cell of thismetal=___Whatis the edge length of a unit cell of Fe=___
The density of solid Fe is 7.87 g/cm3. How many atoms are presentper cubic centimeter of Fe =__
As a solid Fe adopts a body-centered cubic unit cell. How many unitcells are present per cubic centimeter of Fe,=___What is the volume of a unit cell of thismetal=___Whatis the edge length of a unit cell of Fe=___
Trinidad TremblayLv2
28 Sep 2019