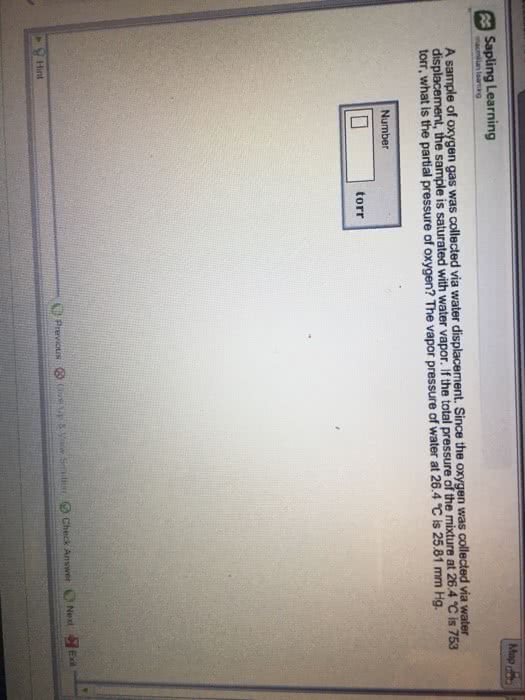
1
answer
0
watching
953
views
15 Nov 2019
Zinc metal is added to hydrochloric acid to generate hydrogen gas and itâs collected over a liquid whose vapor pressure is the same as pure water. A 130 mL sample of gas is collected over water at 22°C and 753 torr. What is the mass of hydrogen that was collected? (the water vapor pressure of water at 22°C = 20. torr)
Zinc metal is added to hydrochloric acid to generate hydrogen gas and itâs collected over a liquid whose vapor pressure is the same as pure water. A 130 mL sample of gas is collected over water at 22°C and 753 torr. What is the mass of hydrogen that was collected? (the water vapor pressure of water at 22°C = 20. torr)
Lelia LubowitzLv2
15 Nov 2019