1
answer
1
watching
743
views
15 Nov 2019
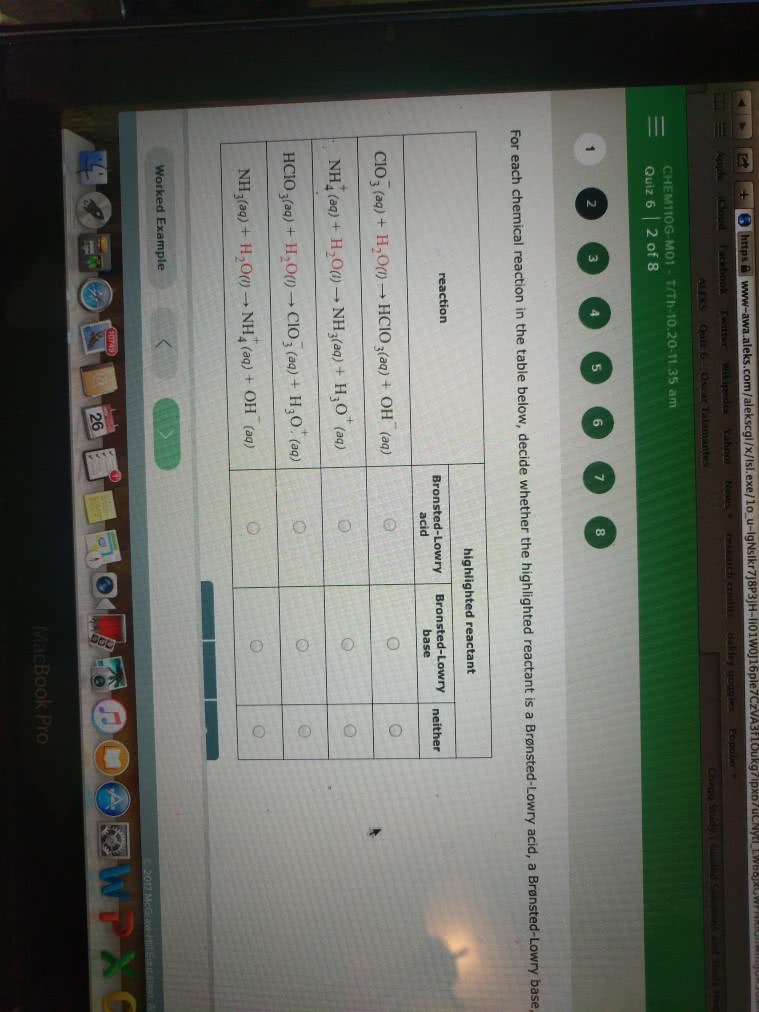
www-awa.aleks.com/alekscgl/x/Isl.exe/1o u-IgNslkr7j8P3JH-li01W0J16ple7CzVA3 loukg CHEM11OG-MO1 T/Th-10.20-11.35 am Quiz 6 | 2 of 8 2 3 5 6 8 For each chemical reaction in the table below, decide whether the highlighted reactant is a Bronsted-Lowry acid, a Brensted highlighted reactant reaction Bronsted-Lowry Bronsted-Lowry neither acid base ClO3 (aq)+ H,00 HCIO3(aa)+ OH () NH4 (aq) + H2O(1) â NH 3(aq) + H30' (aq) Hcio 3(aq) + H20() â CIO 3 (aq) + H30' (aq) NH3(aq) + H2O() â NH4 (aq) + OH-(aq) Worked Example 26

www-awa.aleks.com/alekscgl/x/Isl.exe/1o u-IgNslkr7j8P3JH-li01W0J16ple7CzVA3 loukg CHEM11OG-MO1 T/Th-10.20-11.35 am Quiz 6 | 2 of 8 2 3 5 6 8 For each chemical reaction in the table below, decide whether the highlighted reactant is a Bronsted-Lowry acid, a Brensted highlighted reactant reaction Bronsted-Lowry Bronsted-Lowry neither acid base ClO3 (aq)+ H,00 HCIO3(aa)+ OH () NH4 (aq) + H2O(1) â NH 3(aq) + H30' (aq) Hcio 3(aq) + H20() â CIO 3 (aq) + H30' (aq) NH3(aq) + H2O() â NH4 (aq) + OH-(aq) Worked Example 26
Jarrod RobelLv2
11 Apr 2019
