Write a balanced equation that describes the net ionic reaction used to prepare your buffer in Part I. Identify the two components of the solution that enable it to resist changes in pH.
-----------------------------------------------------------------------------------------------------------------------
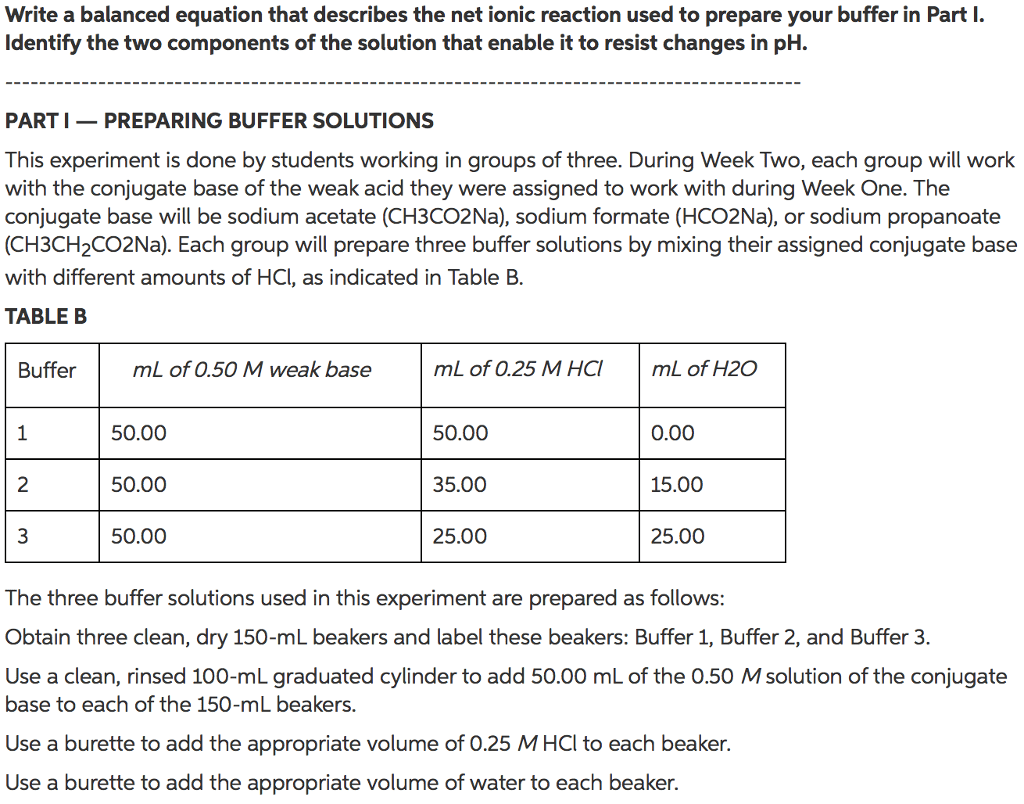
PART I â PREPARING BUFFER SOLUTIONS
This experiment is done by students working in groups of three. During Week One, each group will be assigned one specific weak acid to work with: acetic acid (CH3CO2H), formic acid (HCO2H), or propanoic acid (CH3CH2CO2H). Each group will prepare three buffer solutions by mixing their assigned weak acid with different amounts of NaOH, as indicated in Table A.
TABLE A
Buffer mL of 0.50 M weak acid
mL of 0.25 M NaOH mL of H2O
1 50.00 50.00 0.00 2 50.00 35.00 15.00 3 50.00 25.00 25.00
The three buffer solutions used in this experiment are prepared as follows:
Obtain three clean, dry 150-mL beakers and label these beakers: Buffer 1, Buffer 2, and Buffer 3.
Use a clean, rinsed 100-mL graduated cylinder to add 50.00 mL of the 0.50 M solution of the weak acid to each of the 150-mL beakers.
Use a burette to add the appropriate volume of 0.25 M NaOH to each beaker.
Use a burette to add the appropriate volume of water to each beaker.
Write a balanced equation that describes the net ionic reaction used to prepare your buffer in Part I. Identify the two components of the solution that enable it to resist changes in pH.
-----------------------------------------------------------------------------------------------------------------------
PART I â PREPARING BUFFER SOLUTIONS
This experiment is done by students working in groups of three. During Week One, each group will be assigned one specific weak acid to work with: acetic acid (CH3CO2H), formic acid (HCO2H), or propanoic acid (CH3CH2CO2H). Each group will prepare three buffer solutions by mixing their assigned weak acid with different amounts of NaOH, as indicated in Table A.
TABLE A
| Buffer | mL of 0.50 M weak acid | mL of 0.25 M NaOH |
| |
| 1 | 50.00 | 50.00 | 0.00 | |
| 2 | 50.00 | 35.00 | 15.00 | |
| 3 | 50.00 | 25.00 | 25.00 |
The three buffer solutions used in this experiment are prepared as follows:
Obtain three clean, dry 150-mL beakers and label these beakers: Buffer 1, Buffer 2, and Buffer 3.
Use a clean, rinsed 100-mL graduated cylinder to add 50.00 mL of the 0.50 M solution of the weak acid to each of the 150-mL beakers.
Use a burette to add the appropriate volume of 0.25 M NaOH to each beaker.
Use a burette to add the appropriate volume of water to each beaker.