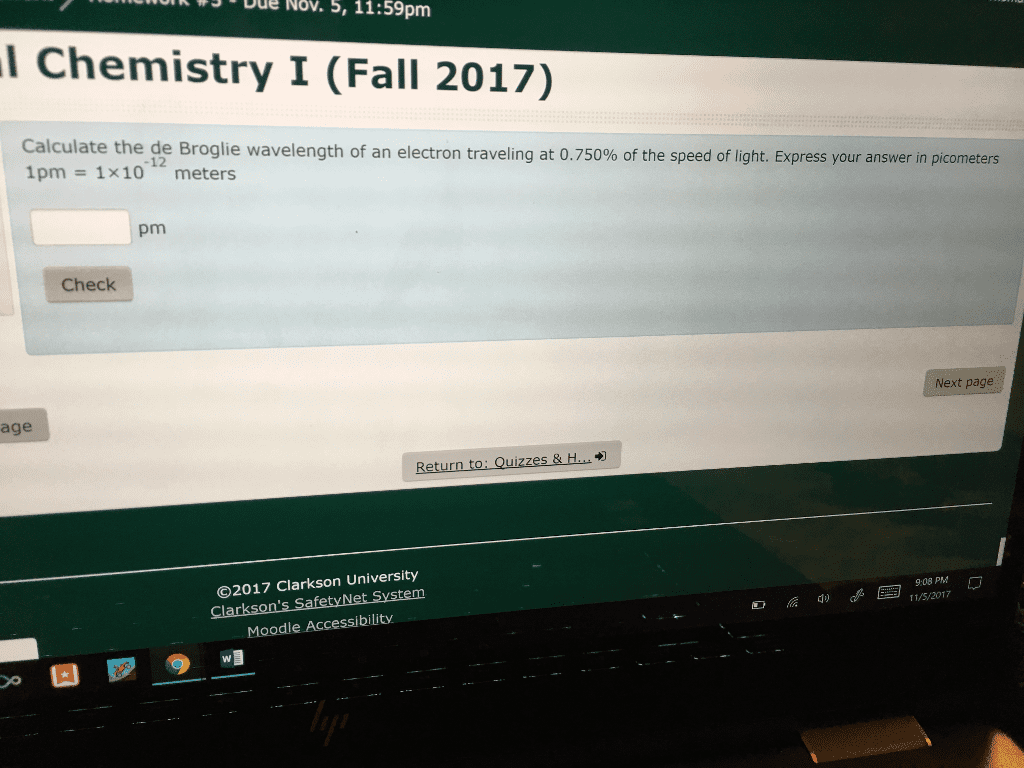
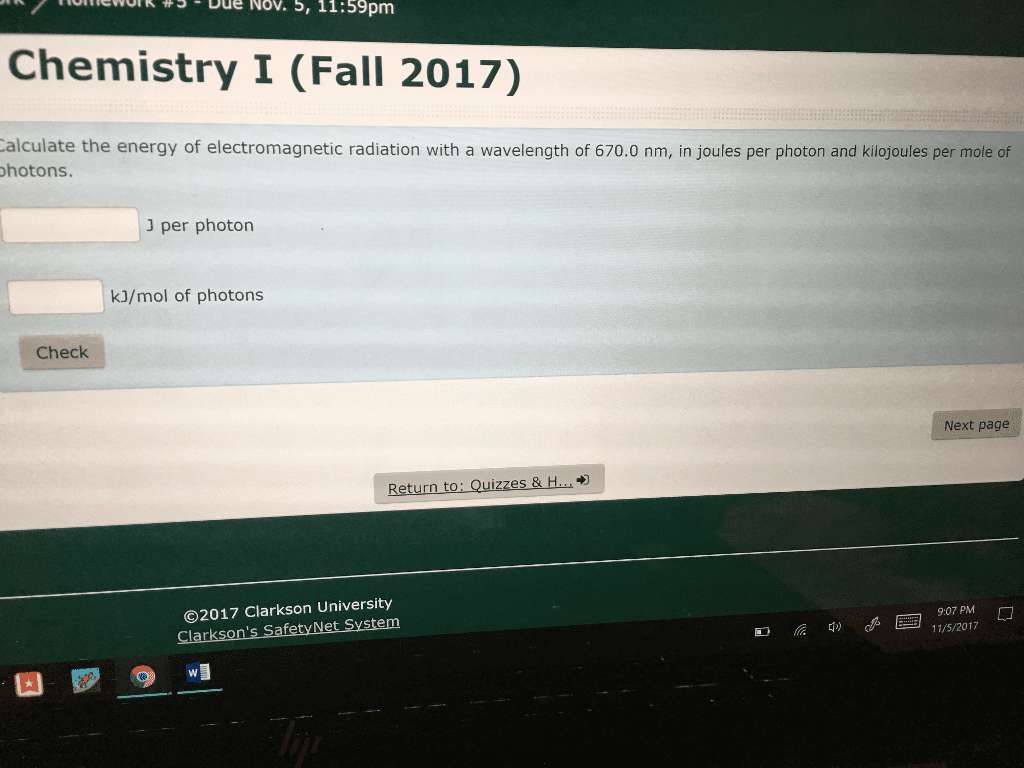
1
answer
0
watching
155
views
15 Nov 2019
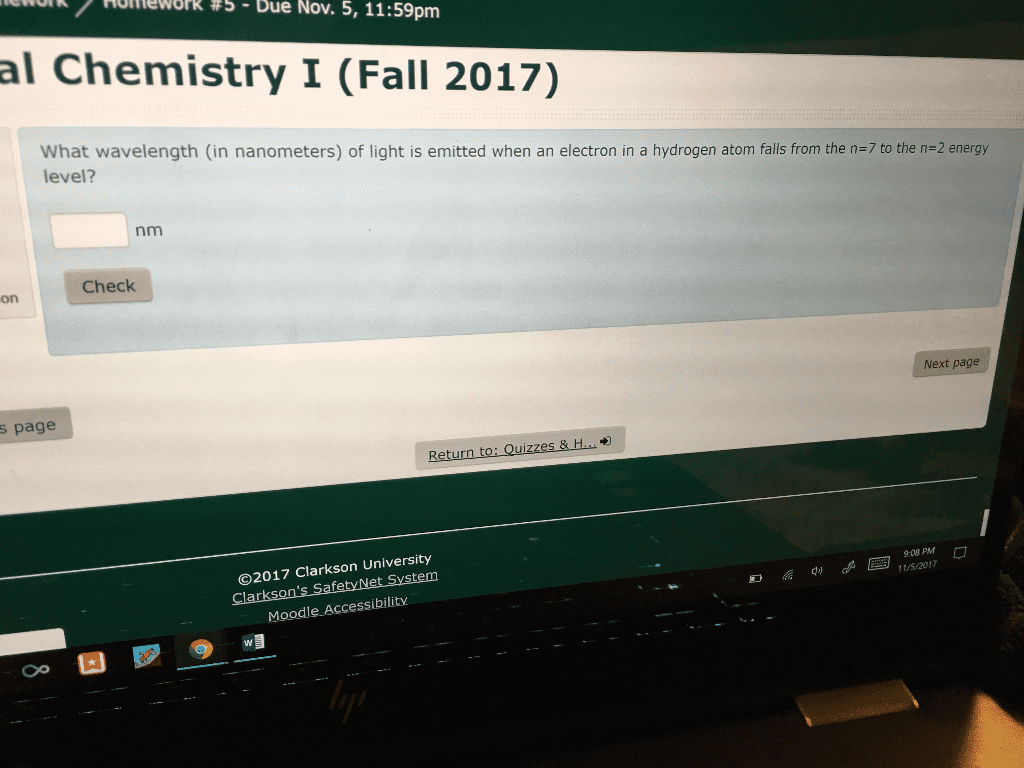
work / Homework #S-Due Nov. 5, 11:59pm al Chemistry I (Fall 2017) What wavelength (in nanometers) of light is emitted when an electron in a hydrogen atom falls from the n-7 to the n 2 energy level? nm Check on Next page s page zzes & H 9:08 PM 15/2017 ©2017 Clarkson University Moodle Accessibilitv

work / Homework #S-Due Nov. 5, 11:59pm al Chemistry I (Fall 2017) What wavelength (in nanometers) of light is emitted when an electron in a hydrogen atom falls from the n-7 to the n 2 energy level? nm Check on Next page s page zzes & H 9:08 PM 15/2017 ©2017 Clarkson University Moodle Accessibilitv
Trinidad TremblayLv2
11 Jan 2019