0
answers
0
watching
210
views
15 Nov 2019


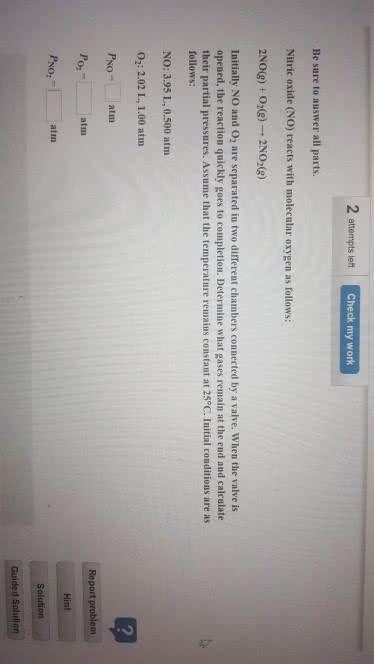
Be sure to answer all parts. Nitroglycerin, an explosive, decomposes according to the following equation 4C3Hs(NOJ3(s)-â 12CO2(g) +10H20(g) + 6N2(g) + O2(g) Caleulate the total volume of gases produced when collected at 1.40 atm, and 24.0°C from 2.93 X 102g of nitroglycerin. What are the partial pressures of the gases under these conditions? atm PH2O atm atm atm 02
0
answers
0
watching
210
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232