0
answers
0
watching
275
views
15 Nov 2019
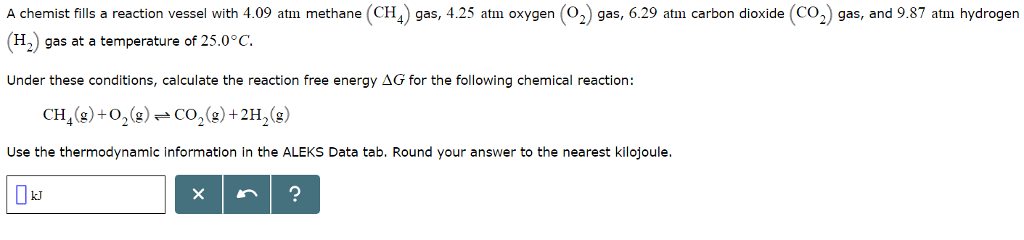
substance Î
Hf °
(kJ/mol) Î
Gf °
(kJ/mol) S °
(J/molâK) Aluminum Al3+ (aq) --- -485.0 --- Al (s) 0 0 28.3 Al2O3 (s) -1675.7 -1582.3 50.9 Al(OH)3 (s) --- -1147.25 --- Bromine Brâ (aq) --- -104.0 --- Br2 (l) 0 0 152.2 Br2 (g) 30.9 3.1 245.5 HBr (g) -36.3 -53.4 198.7 Calcium Ca2+ (aq) --- -553.6 --- CaCl2 (s) -795.4 --- --- CaO (s) -634.9 --- --- Ca(OH)2 (s) -985.2 --- --- Ca3(PO4)2 (s) --- -3884.7 --- Carbon C (s) 0 0 5.7 CCl4 (g) -95.7 -64.0 309.4 CCl4 (l) -128.2 -68.6 214.4 CCl4 (s) -136.8 --- --- CH4 (g) -74.6 -50.5 186.3 C2H2 (g) 227.4 --- --- C2H6 (g) -84.0 --- --- C3H8 (g) -103.85 -23.4 270.3 C4H10 (g) -125.7 -15.7 310.0 C4H10 (l) -147.3 -15.0 231.0 C6H6 (l) 49.1 124.5 173.4 C6H6 (g) 82.9 129.7 269.2 CH3OH (l) -239.2 -166.6 126.80 CH3OH (g) -201.0 -162.3 239.9 (CH3)2(CH2)2O (l) -279.5 -346.74 253.5 C6H12O6 (s) -1237.3 -910.4 212.1 CO (g) -110.5 -137.2 197.7 CO2 (g) -393.5 -394.4 213.8 C2H5OH (l) -277.6 -174.8 160.7 C2H5OH (g) -234.8 -167.9 281.6 HCH3CO2 (l) -484.3 -389.9 159.8 HCH3CO2 (s) -472.8 --- --- HCH3CO2 (g) -432.2 --- 283.5 HCN (l) 108.9 125.0 112.8 HCN (g) 135.1 124.7 201.8 CS2 (s) 93.8 --- --- CS2 (l) 89.0 64.6 151.3 CS2 (g) 116.7 67.1 237.8 Chlorine Cl2 (g) 0 0 223.1 Clâ (aq) --- -131.2 --- HCl (aq) -167. --- --- HCl (g) -92.3 -95.3 186.9 Chromium CrO42â (aq) --- -727.8 --- Fluorine F2 (g) 0 0 202.8 HF (g) -273.3 -275.4 173.8 Hydrogen H2 (g) 0 0 130.7 Iron Fe (s) 0 0 27.3 FeCl3 (s) -399.5 -334.0 142.3 Fe2O3 (s) -824.2 -742.2 87.4 Lead Pb2+ (aq) --- -24.4 --- PbBr2 (s) -278.7 -261.9 161.5 PbS (s) -100.4 -98.7 91.2 PbSO4 (s) -920.0 -813.0 148.5 Magnesium MgCl2 (s) -641.3 -591.8 89.6 MgO (s) -601.6 -569.3 27.0 Mercury Hg (g) 61.4 31.8 175.0 Hg (l) 0 0 75.9 Hg (s) -2.29 --- --- Hg22+ (aq) --- 153.5 --- Hg2Cl2 (s) --- -210.7 --- Nitrogen N2 (g) 0 0 191.6 N2H4 (g) 95.4 159.4 238.5 N2H4 (l) 50.6 149.3 121.2 NH3 (g) -45.9 -16.4 192.8 NH3 (l) -69.54 -26.50 95.09 NH4NO3 (s) -365.6 -183.9 151.1 NO (g) 91.3 87.6 210.8 NO2 (g) 33.2 51.3 240.1 NOCl (g) 51.7 66.1 261.7 N2O4 (g) 11.1 99.8 304.4 HNO2 (g) -79.5 --- --- HNO3 (l) -174.1 -80.7 155.6 HNO3 (g) -133.9 -73.5 266.9 Oxygen OHâ (aq) --- -157.2 --- H2O (l) -285.8 -237.1 70.0 H2O (g) -241.8 -228.6 188.8 O2 (g) 0 0 205.2 H2O2 (l) -187.8 -120.4 109.6 H2O2 (g) -136.3 -105.6 232.7 Phosphorus P4 (s) 0 0 41. P4 (g) 58.9 24.4 280.0 PCl3 (g) -287.0 -267.8 311.8 PCl3 (l) -319.7 -272.3 217.1 PF5 (g) -1594.4 -1520.7 300.8 PO43â (aq) --- -1018.7 --- H3PO4 (l) -1271.7 --- --- H3PO4 (s) -1284.4 -1124.3 110.5 P4O10 (s) -2984. -2675.2 228.9 Potassium KBr (s) -393.4 -380.7 95.9 KNO3 (s) -492.7 -393.13 132.9 Silver Ag+ (aq) --- 77.1 --- Ag2CrO4 (s) --- -641.8 --- Sodium NaCl (s) -411.2 --- --- NaOH (s) -425.8 --- --- Sulfur S8 (s) 0 0 32.0 H2S (g) -20.6 --- --- H2SO4 (l) -814.0 --- --- SF6 (g) -1220.5 -1116.5 291.5 Tin Sn (s) 0 0 51.2 SnO2 (s) -577.6 -515.8 49.0 Titanium TiCl4 (g) -763.2 -726.3 353.2 TiCl4 (l) -804.2 -737.2 252.3 TiO2 (s) -944.0 -888.8 50.6
A chemist fills a reaction vessel with 4.09 atm methane (CH gas, 4.25 atm oxygen (2) gas, 6.29 atm carbon dioxide (CO2 gas, and 9.87 atm hydrogen (H2) gas at a temperature of 25.0° C. Under these conditions, calculate the reaction free energy ÎG for the following chemical reaction: CH4(g) +02(g)--CO2(g)+2H2(g) Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A chemist fills a reaction vessel with 4.09 atm methane (CH gas, 4.25 atm oxygen (2) gas, 6.29 atm carbon dioxide (CO2 gas, and 9.87 atm hydrogen (H2) gas at a temperature of 25.0° C. Under these conditions, calculate the reaction free energy ÎG for the following chemical reaction: CH4(g) +02(g)--CO2(g)+2H2(g) Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule
