0
answers
0
watching
125
views
15 Nov 2019


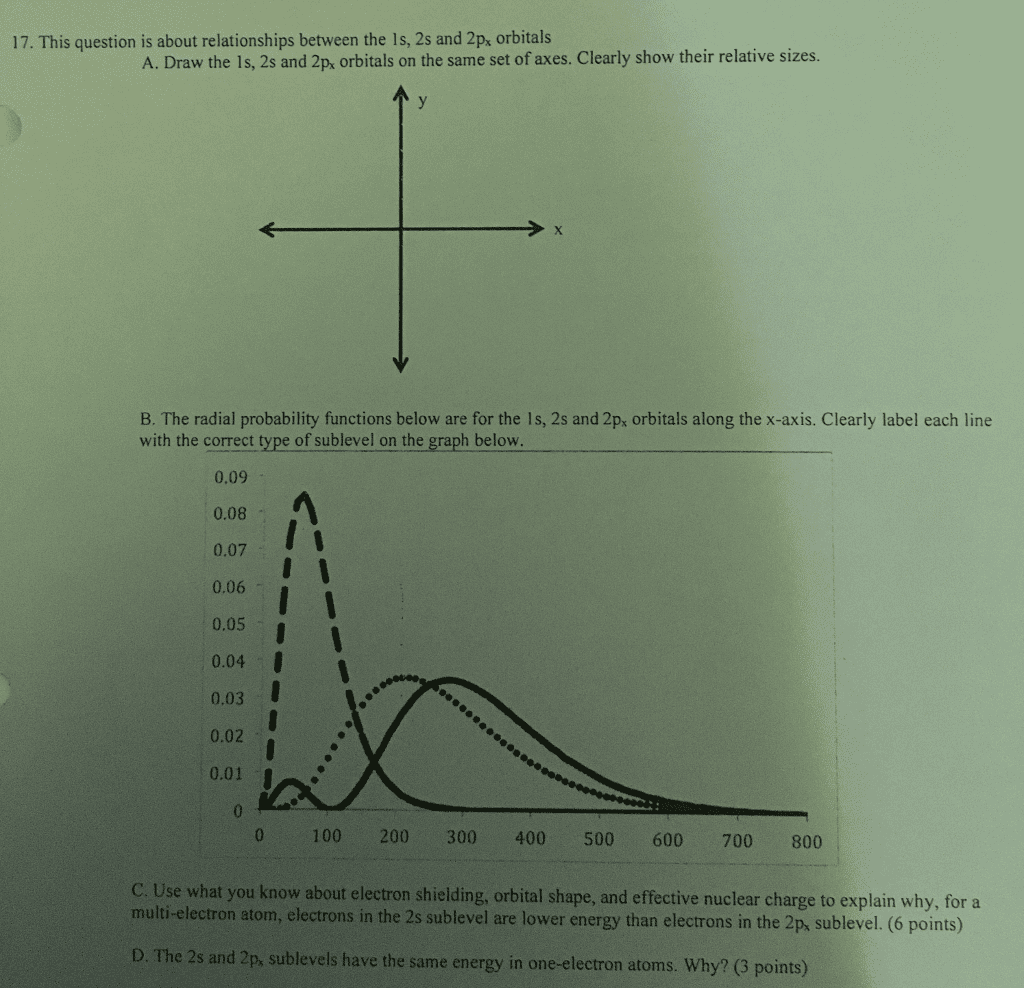
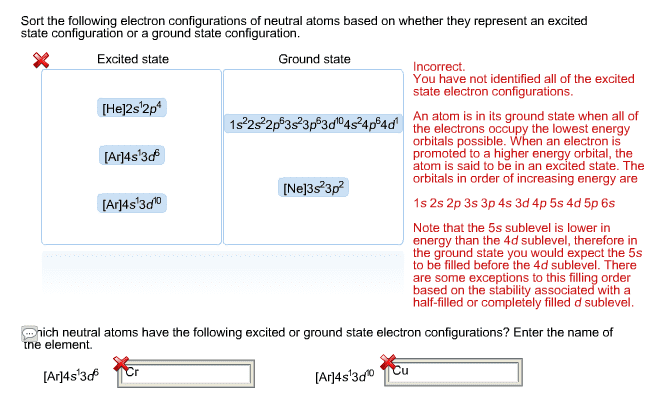
Submit My Answers Give Up Correct Part B If we add one electron to form the He atom, would your answer to part (a) change? O Yes. In an He atom, the 2s orbital is lower in energy than the 2p orbitals O No, the 2s orbital of He has the greater energy O No, the 2p orbital of He has the greater energy O cannot be determined Submit My Answers Give Up
0
answers
0
watching
125
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232