2
answers
0
watching
313
views
15 Nov 2019
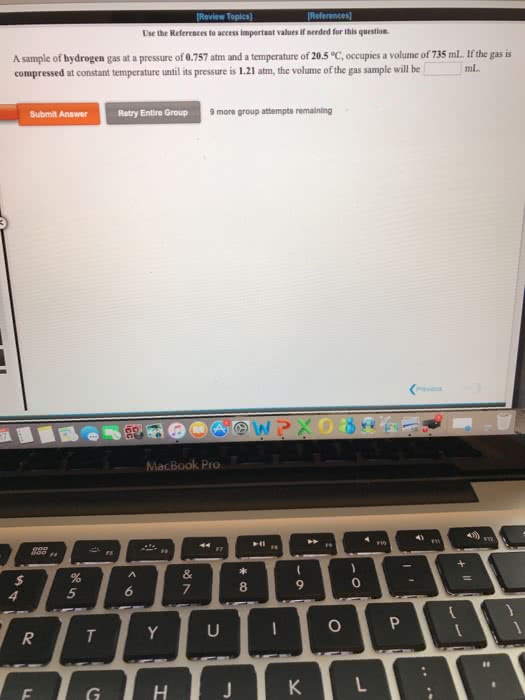
A sample of methane gas at a pressure of 1.09 atm and a temperature of 20.8 °C, occupies a volume of 14.3 liters. If the gas is compressed at constant temperature to a volume of 5.73 liters, the pressure of the gas sample will be in atm.
A sample of oxygen gas at a pressure of 0.919 atm and a temperature of 29.7 °C, occupies a volume of 711 mL. If the gas is allowed to expand at constant temperature until its pressure is 0.673 atm, the volume of the gas sample will be in mL.
A sample of helium gas at a pressure of 0.820 atm and a temperature of 23.4 °C, occupies a volume of 845 mL. If the gas is compressed at constant temperature until its pressure is 1.28 atm, the volume of the gas sample will be in mL
A sample of methane gas at a pressure of 1.09 atm and a temperature of 20.8 °C, occupies a volume of 14.3 liters. If the gas is compressed at constant temperature to a volume of 5.73 liters, the pressure of the gas sample will be in atm.
A sample of oxygen gas at a pressure of 0.919 atm and a temperature of 29.7 °C, occupies a volume of 711 mL. If the gas is allowed to expand at constant temperature until its pressure is 0.673 atm, the volume of the gas sample will be in mL.
A sample of helium gas at a pressure of 0.820 atm and a temperature of 23.4 °C, occupies a volume of 845 mL. If the gas is compressed at constant temperature until its pressure is 1.28 atm, the volume of the gas sample will be in mL
thanhmai1309Lv9
31 Aug 2023
Deanna HettingerLv2
21 Sep 2019
Already have an account? Log in