1
answer
0
watching
197
views
16 Nov 2019
For the reaction shown:
1. Calculate the enthalpy change.
2. Assuming the entropy change is zero, calculate the equilibrium constant at 298 K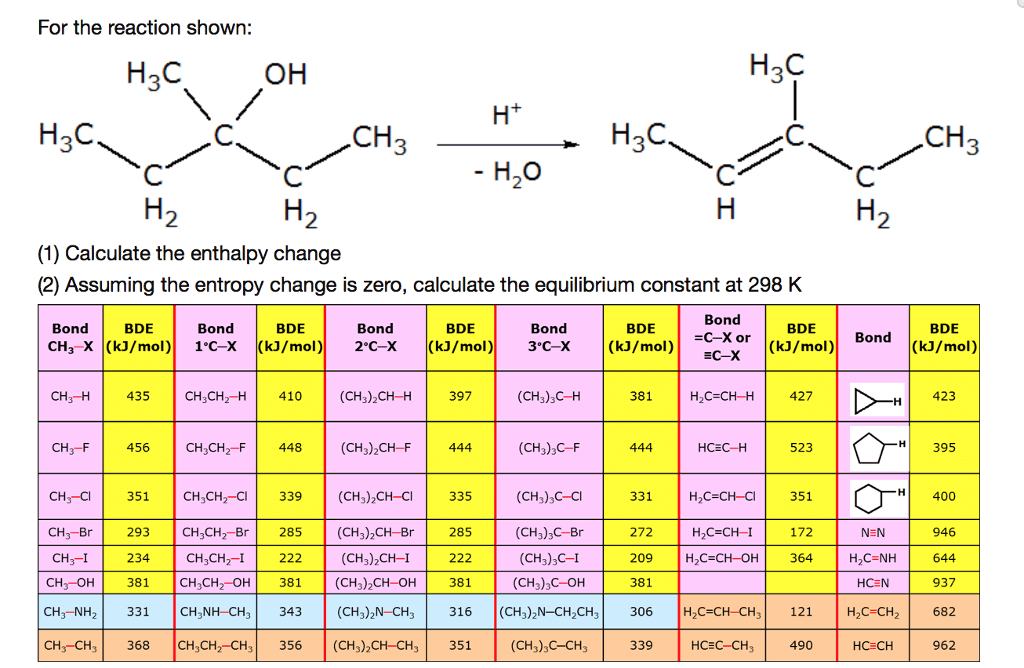
For the reaction shown: H2C OH H2C H2C CH H2C CH 3 2 (1) Calculate the enthalpy change (2) Assuming the entropy change is zero, calculate the equilibrium constant at 298 K Bond BDE EC-x (kJ/mol) Bond Bond BDE Bond BDE Bond BDE Bond BDE | BDE | =C-X or CH3-X (kJ/mol)1°C-X (kJ/mol)2°C-X (kJ/mol)3°C-X (kJ/mol) CH3-H435 CH3CH2-H 410 (CH3)2CH-H 397 (CH3)3C-H 381 H2C=CH-H 427 423 CH3-F 456CH3CH2F 448 (CH3)2CH- 444 (CH3)3C-F 523 395 331 351 H 400 CHåCl | 351 CH3CH2-Cl| 339 | (CH3)2CH-Cl | 335 CH3-Br | 293 CH3CH2-Br| 285 (CH3)2CH. Br | 285 CH3-I CH3-OH! 38 1 CH3CH2-OH | 38 1 (CH3)2CH-OH| 381 CH3-NH2 331 CH3NH-CH3343 (CH3)2N-CH3 CH3-CH3 368 CH3CH2-CH3 356 (CH3)2CH-CH3 351 H2C-CH-C (CH3)zC-c (CH3) C-Br (CH3)sC-I (CH3)3C-OH 316 (CH,)2N-CH,CH, 306 272 H2C-CH- 172 209 381 NEN H2C NH HCEN H2C-CH2 HCECH 946 644 937 682 962 234 CH3CH2- 222 (CH3) CH-I H,C-CH-O 364 H2C-CH-CH 121 (CH3),C-CHs 339 HCEC-CH3 490
For the reaction shown:
1. Calculate the enthalpy change.
2. Assuming the entropy change is zero, calculate the equilibrium constant at 298 K
For the reaction shown: H2C OH H2C H2C CH H2C CH 3 2 (1) Calculate the enthalpy change (2) Assuming the entropy change is zero, calculate the equilibrium constant at 298 K Bond BDE EC-x (kJ/mol) Bond Bond BDE Bond BDE Bond BDE Bond BDE | BDE | =C-X or CH3-X (kJ/mol)1°C-X (kJ/mol)2°C-X (kJ/mol)3°C-X (kJ/mol) CH3-H435 CH3CH2-H 410 (CH3)2CH-H 397 (CH3)3C-H 381 H2C=CH-H 427 423 CH3-F 456CH3CH2F 448 (CH3)2CH- 444 (CH3)3C-F 523 395 331 351 H 400 CHåCl | 351 CH3CH2-Cl| 339 | (CH3)2CH-Cl | 335 CH3-Br | 293 CH3CH2-Br| 285 (CH3)2CH. Br | 285 CH3-I CH3-OH! 38 1 CH3CH2-OH | 38 1 (CH3)2CH-OH| 381 CH3-NH2 331 CH3NH-CH3343 (CH3)2N-CH3 CH3-CH3 368 CH3CH2-CH3 356 (CH3)2CH-CH3 351 H2C-CH-C (CH3)zC-c (CH3) C-Br (CH3)sC-I (CH3)3C-OH 316 (CH,)2N-CH,CH, 306 272 H2C-CH- 172 209 381 NEN H2C NH HCEN H2C-CH2 HCECH 946 644 937 682 962 234 CH3CH2- 222 (CH3) CH-I H,C-CH-O 364 H2C-CH-CH 121 (CH3),C-CHs 339 HCEC-CH3 490
Collen VonLv2
31 May 2019


