2
answers
1
watching
973
views
greenmole103Lv1
28 Sep 2019
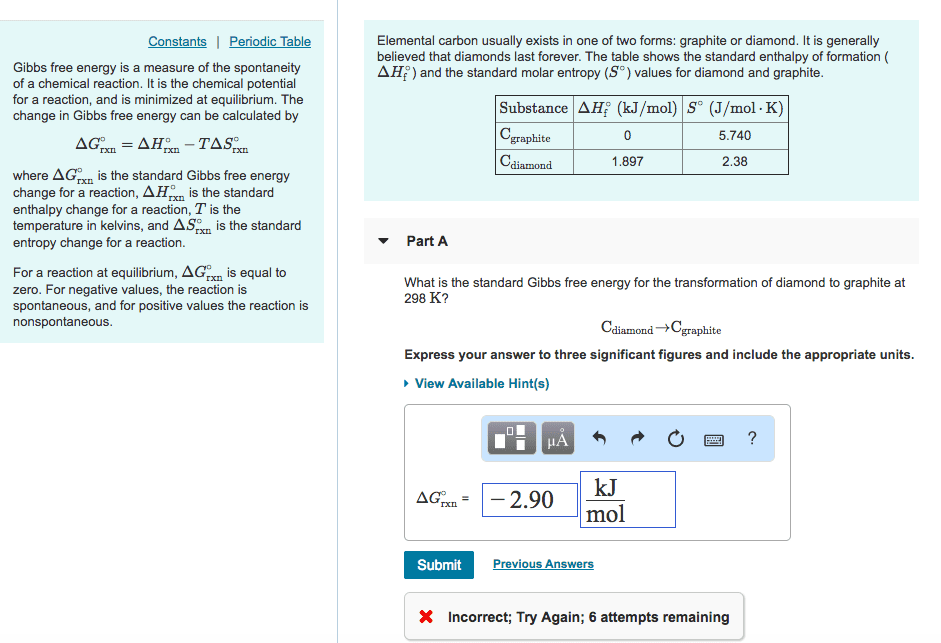
Elemental carbon usually exists in one of two forms: graphite or diamond. It is generally believed that diamonds last forever. The table shows the standard enthalpy of formation (?H?f) and the standard molar entropy (S?) values for diamond and graphite.
Substance ?H?f (kJ/mol) S? (J/mol?K) Cgraphite 0 5.740 Cdiamond 1.897 2.38
What is the standard Gibbs free energy for the transformation of diamond to graphite at 298 K?
Cdiamond?Cgraphite
Express your answer to three significant figures and include the appropriate units.
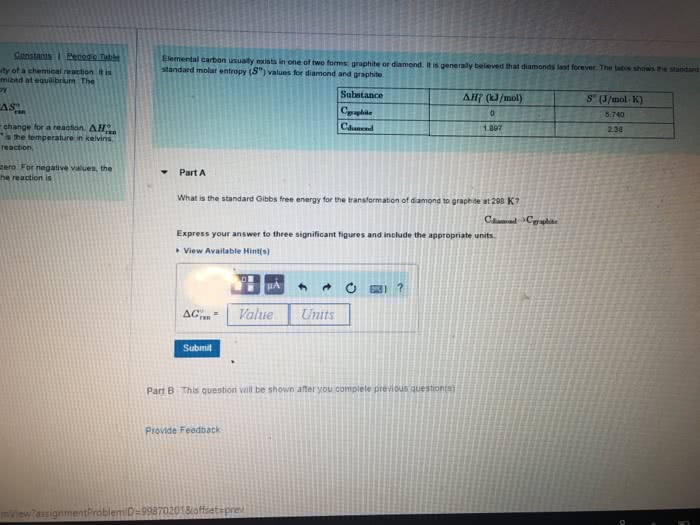
Elemental carbon usually exists in one of two forms: graphite or diamond. It is generally believed that diamonds last forever. The table shows the standard enthalpy of formation (?H?f) and the standard molar entropy (S?) values for diamond and graphite.
| Substance | ?H?f (kJ/mol) | S? (J/mol?K) |
| Cgraphite | 0 | 5.740 |
| Cdiamond | 1.897 | 2.38 |
What is the standard Gibbs free energy for the transformation of diamond to graphite at 298 K?
Cdiamond?Cgraphite
Express your answer to three significant figures and include the appropriate units.
Liked by samareshj1123
Hubert KochLv2
28 Sep 2019
16 Mar 2024
Already have an account? Log in