2
answers
0
watching
765
views
ivorywasp158Lv1
28 Sep 2019
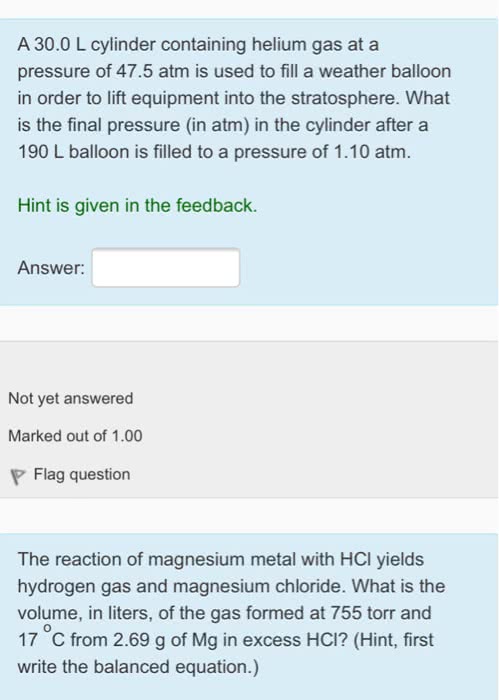
1. A 20 L cylinder containing helium gas at a pressure of 29.5 atmis used to fill a weather balloon in order to lift equipment intothe stratosphere. To what maximum pressure (in atm) could a 155 Lballoon be filled.
2. A 27.0L cylinder containing helium gas at a pressure of 43.0 atm is usedto fill a weather balloon in order to lift equipment into thestratosphere. What is the final pressure (in atm) in the cylinderafter a 219 L balloon is filled to a pressure of 1.25atm.
3.Thereaction of magnesium metal with HCl yields hydrogen gas andmagnesium chloride. What is the volume, in liters, of the gasformed at 760 torr and 21 oC from 6.99 g of Mg in excessHCl? (Hint, first write the balanced equation.)
1. A 20 L cylinder containing helium gas at a pressure of 29.5 atmis used to fill a weather balloon in order to lift equipment intothe stratosphere. To what maximum pressure (in atm) could a 155 Lballoon be filled.
2. A 27.0L cylinder containing helium gas at a pressure of 43.0 atm is usedto fill a weather balloon in order to lift equipment into thestratosphere. What is the final pressure (in atm) in the cylinderafter a 219 L balloon is filled to a pressure of 1.25atm.
3.Thereaction of magnesium metal with HCl yields hydrogen gas andmagnesium chloride. What is the volume, in liters, of the gasformed at 760 torr and 21 oC from 6.99 g of Mg in excessHCl? (Hint, first write the balanced equation.)
28 Jul 2023
Jamar FerryLv2
28 Sep 2019
Already have an account? Log in