1
answer
0
watching
1,038
views
28 Sep 2019
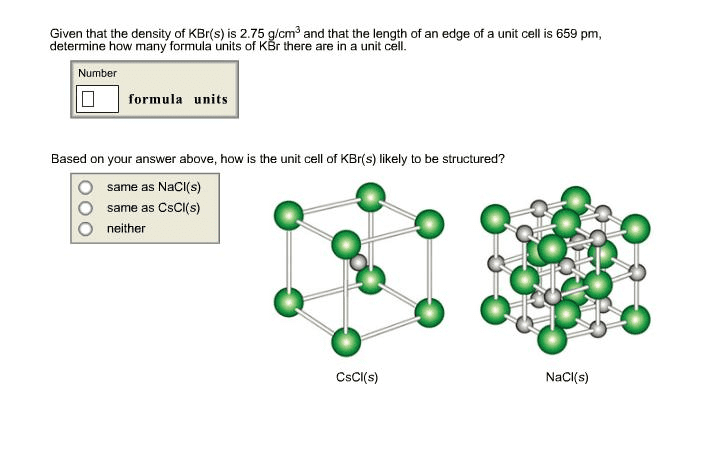
Given that the density of CaO(s) is 3.34 g/cm3 and that the lengthof an edge of a unit cell is 481 pm, determine how many formulaunits of CaO there are in a unit cell.
The only answer i need is the one that is in formulaunits.
Given that the density of CaO(s) is 3.34 g/cm3 and that the lengthof an edge of a unit cell is 481 pm, determine how many formulaunits of CaO there are in a unit cell.
The only answer i need is the one that is in formulaunits.
Jean KeelingLv2
28 Sep 2019