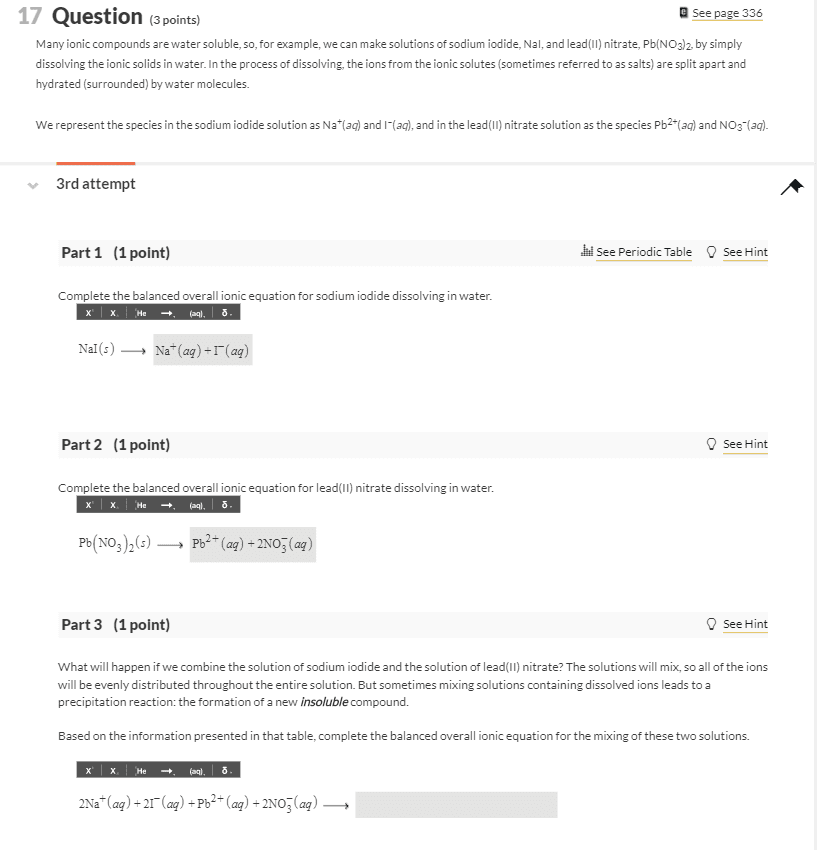
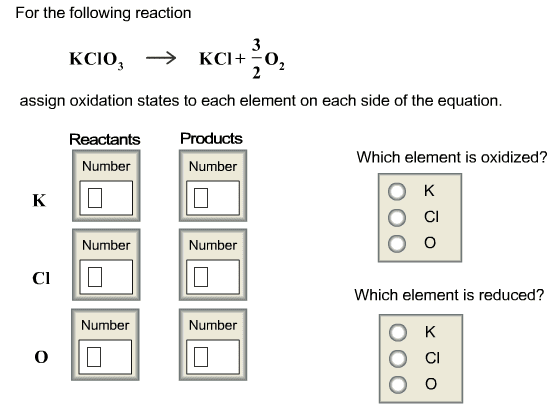
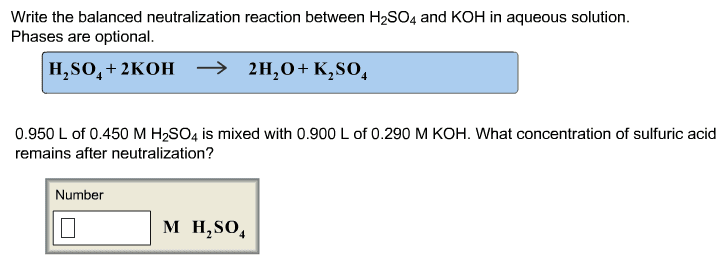
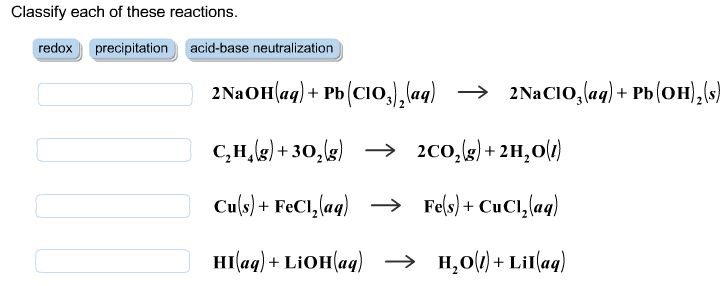4. Solubility Rules and Precipitation Reactions. Do both problems .4A Imagine you have a solution containing a mixture of Mg2+(aq), Ba2+ (aq), Ag+ (aq) and NO3 - (aq) dissolved in the solution. You have available to you 5 different solutions containing the ions shown: Solution 1. Na+ (aq) and Cl- (aq) Solution 2. Na+ (aq) and OH- (aq) Solution 3. Na+ (aq) and SO4 2- (aq) Solution 4. Na+ (aq) and PO4 3- (aq) Solution 5. Na+ (aq) and NO3 - (aq) a. Which of the five solutions could you use to remove the Ba2+ as an insoluble solid, but leave the Mg2+ and the Ag+ dissolved in solution? Explain why you decided on your answer. b. Which of the five solutions could you use to remove the Mg2+ and the Ag+ as insoluble solids, but leave the Ba2+ dissolved in solution? Explain why you decided on your answer. 4B. Phosphoric acid, H3PO4 is added to aluminum sulfate, Al2(SO4)3 in water. The products are sulfuric acid, H2SO4, and aluminum phosphote, AlPO4. Write and balance this reaction, and use the table of solubility to determine the physical states of all of the reactants and products.
4. Solubility Rules and Precipitation Reactions. Do both problems .4A Imagine you have a solution containing a mixture of Mg2+(aq), Ba2+ (aq), Ag+ (aq) and NO3 - (aq) dissolved in the solution. You have available to you 5 different solutions containing the ions shown: Solution 1. Na+ (aq) and Cl- (aq) Solution 2. Na+ (aq) and OH- (aq) Solution 3. Na+ (aq) and SO4 2- (aq) Solution 4. Na+ (aq) and PO4 3- (aq) Solution 5. Na+ (aq) and NO3 - (aq) a. Which of the five solutions could you use to remove the Ba2+ as an insoluble solid, but leave the Mg2+ and the Ag+ dissolved in solution? Explain why you decided on your answer. b. Which of the five solutions could you use to remove the Mg2+ and the Ag+ as insoluble solids, but leave the Ba2+ dissolved in solution? Explain why you decided on your answer. 4B. Phosphoric acid, H3PO4 is added to aluminum sulfate, Al2(SO4)3 in water. The products are sulfuric acid, H2SO4, and aluminum phosphote, AlPO4. Write and balance this reaction, and use the table of solubility to determine the physical states of all of the reactants and products.