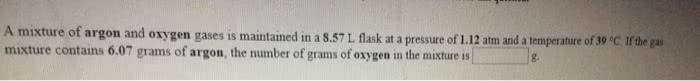1
answer
0
watching
170
views
16 Nov 2019
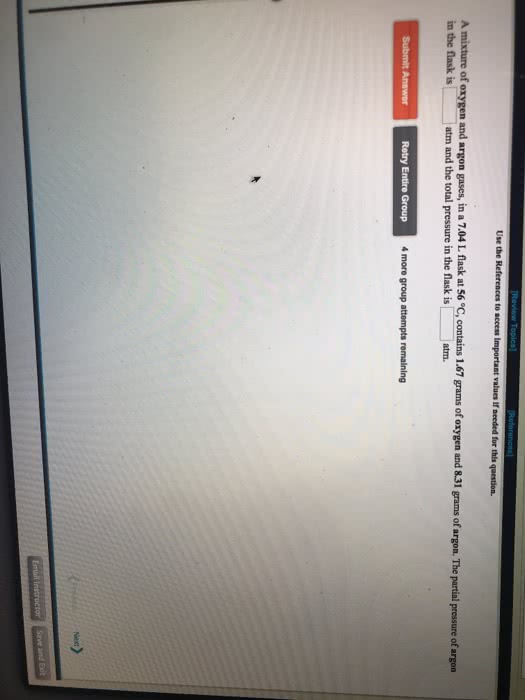
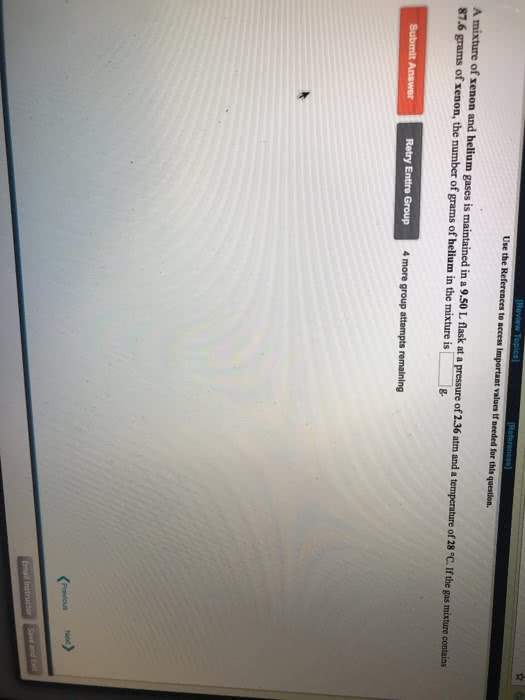
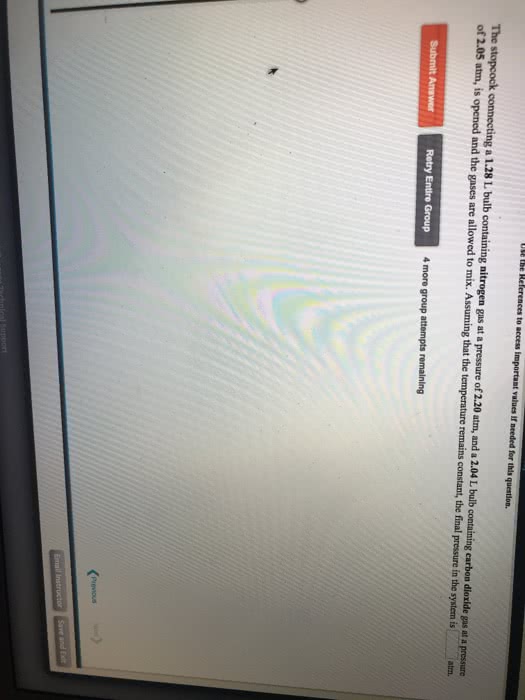
A mixture of argon and oxygen gases is maintained in a 7.83 L flask at a pressure of 2.38 atm and a temperature of 68.0 °C. If the gas mixture contains 8.36 grams of argon, the number of grams of oxygen in the mixture is
A mixture of argon and oxygen gases is maintained in a 7.83 L flask at a pressure of 2.38 atm and a temperature of 68.0 °C. If the gas mixture contains 8.36 grams of argon, the number of grams of oxygen in the mixture is
Keith LeannonLv2
2 Sep 2019