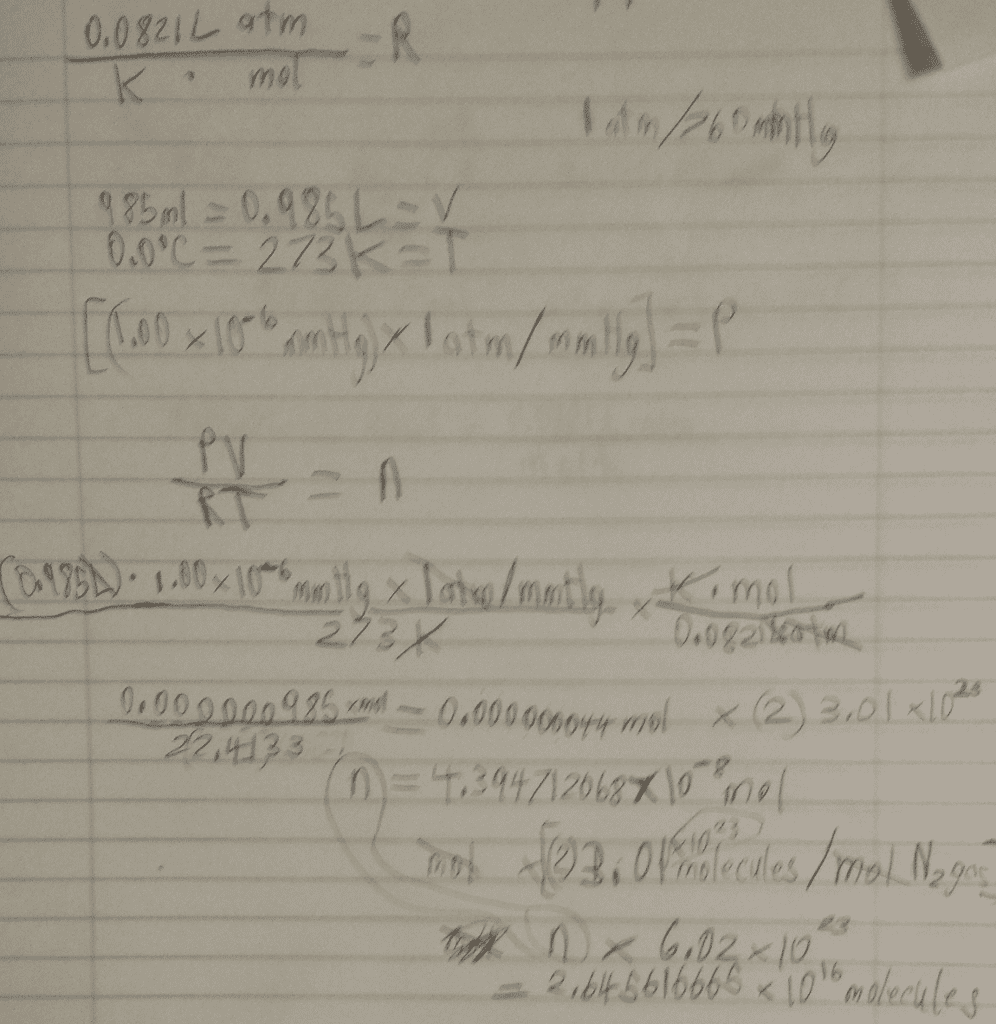what is the meaning of the slope of the line of a graph where P is on the y-axis and 1\V is on the x-axis? Can you figure out how many moles of gas where trapped in the syringe? Assume that the amount of gas (n) and tempreture are fixed. Hint: Manipulate PV=nRT to lool like y=mx. Remeber that y is P and x is 1|v
what is the meaning of the slope of the line of a graph where P is on the y-axis and 1\V is on the x-axis? Can you figure out how many moles of gas where trapped in the syringe? Assume that the amount of gas (n) and tempreture are fixed. Hint: Manipulate PV=nRT to lool like y=mx. Remeber that y is P and x is 1|v
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
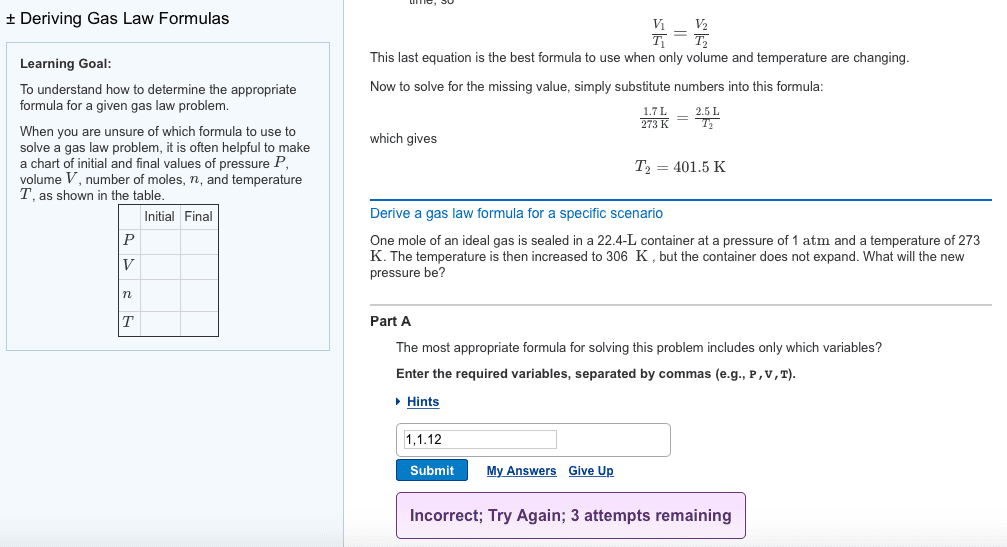
| 1. If 3.9 moles of Mg were mixed with HCl, how many moles of H2 gas would you expect to be produced? 2. If the ambient pressure is 0.991 atm and the temperature is 298 K, what volume (in liters) would moles of H2 occupy? 3. Write out the ideal gas law, and rearrange the equation to solve for volume (V) 4. For a plot of V vs. number of moles (n), where V is on the y-axis and n is on the x-axis, write an expression for the slope in terms of pressure, temperature and R. | | ||||||||