2
answers
0
watching
1,435
views
28 Sep 2019
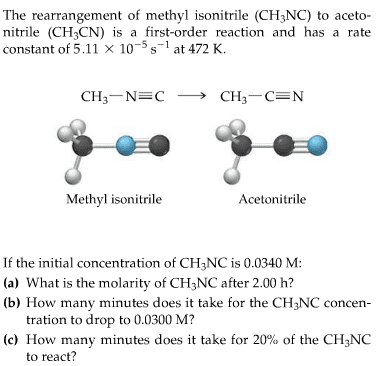
The conversion of methyl isonitrile to acetonitrile in the gas phase at 250 °C CH3NC(g)CH3CN(g) is first order in CH3NC. During one experiment it was found that when the initial concentration of CH3NC was 6.20Ã10-2 M, the concentration of CH3NC dropped to 1.13Ã10-2 M after 433 s had passed. Based on this experiment, the rate constant for the reaction is s-1.
The conversion of methyl isonitrile to acetonitrile in the gas phase at 250 °C CH3NC(g)CH3CN(g) is first order in CH3NC. During one experiment it was found that when the initial concentration of CH3NC was 6.20Ã10-2 M, the concentration of CH3NC dropped to 1.13Ã10-2 M after 433 s had passed. Based on this experiment, the rate constant for the reaction is s-1.
raj21chaharLv10
20 May 2023
Deanna HettingerLv2
28 Sep 2019
Already have an account? Log in