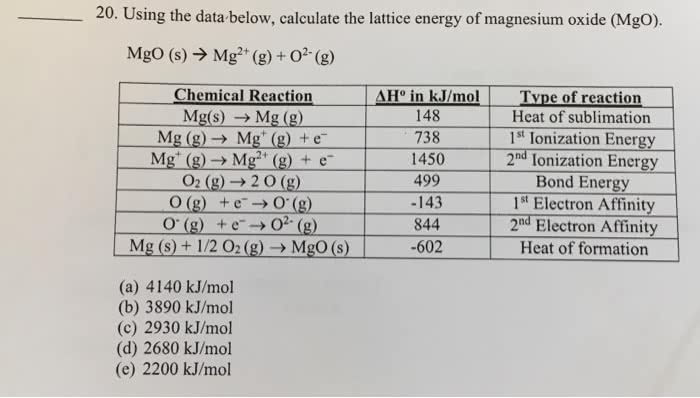
3
answers
0
watching
79
views
28 Sep 2019
Problem: Calculate the ?H2nd electron affinity for Ca usingthe following data for the formation of CaO: Given: ? H sublimation: +192 KJ ? H ionization: the first and second ionization energies are:590.0KJ/mole and 1145. KJ/mole, respectively ? H dissociation: +494 KJ (Bond energy) ? H 1st electron affinity: -141 KJ/mole ? H formation: -636 KJ ? H lattice energy: -3514 The answer is said to be 845 kJ. I'm completely lost inthe steps. Please help! Thanks!
Problem: Calculate the ?H2nd electron affinity for Ca usingthe following data for the formation of CaO:
Given:
? H sublimation: +192 KJ
? H ionization: the first and second ionization energies are:590.0KJ/mole
and 1145. KJ/mole, respectively
? H dissociation: +494 KJ (Bond energy)
? H 1st electron affinity: -141 KJ/mole
? H formation: -636 KJ
? H lattice energy: -3514
The answer is said to be 845 kJ. I'm completely lost inthe steps. Please help! Thanks!
25 Feb 2023
Already have an account? Log in
Nestor RutherfordLv2
28 Sep 2019
Already have an account? Log in