The molar heat capacity C P,m of SO 2 (g) is described by the following equation over the range 300 K < T < 1700 K :
C P,m R = 3.093+6.967Ã10^â3 T/K
â45.81Ã10^â7 T^2/K^2 +1.035Ã10^â9 T^3/K^3
In this equation, T is the absolute temperature in kelvin. The ratios T^n /K^n ensure that C P,m has the correct dimension. 1.40 moles of SO 2 (g) is heated from 28.0 â C to 1.10Ã103 â C at a constant pressure of 1 bar . Solve for calculate ÎH,q ,ÎU and w.
The molar heat capacity C P,m of SO 2 (g) is described by the following equation over the range 300 K < T < 1700 K :
C P,m R = 3.093+6.967Ã10^â3 T/K
â45.81Ã10^â7 T^2/K^2 +1.035Ã10^â9 T^3/K^3
In this equation, T is the absolute temperature in kelvin. The ratios T^n /K^n ensure that C P,m has the correct dimension. 1.40 moles of SO 2 (g) is heated from 28.0 â C to 1.10Ã103 â C at a constant pressure of 1 bar . Solve for calculate ÎH,q ,ÎU and w.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
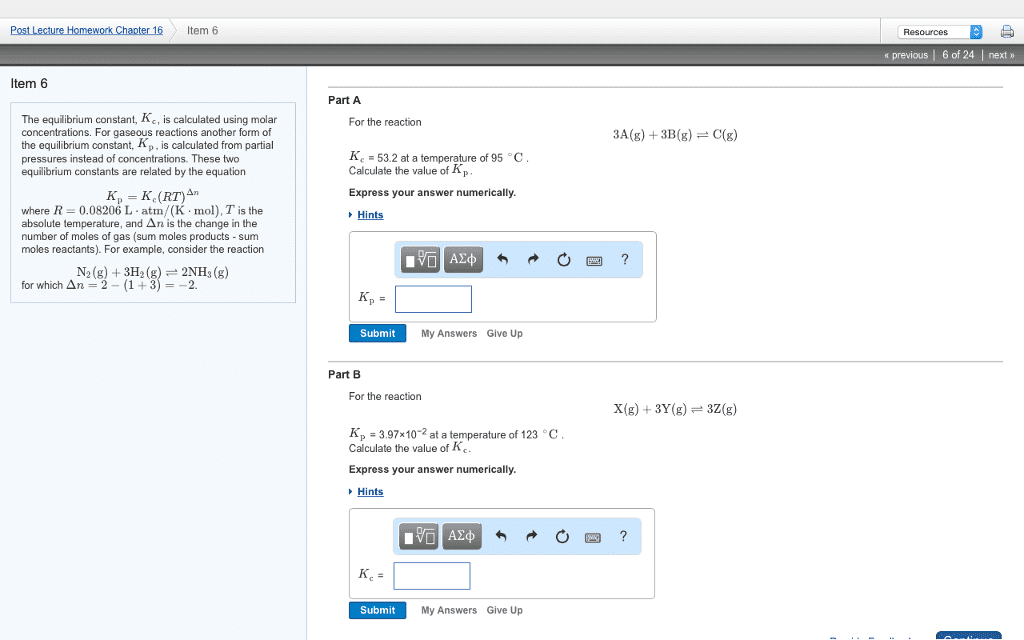
| The equilibrium constant, Kc, is calculated using molar concentrations. For gaseous reactions another form of the equilibrium constant, Kp, is calculated from partial pressures instead of concentrations. These two equilibrium constants are related by the equation Kp=Kc(RT)În where R=0.08206 Lâ atm/(Kâ mol), T is the absolute temperature, and În is the change in the number of moles of gas (sum moles products - sum moles reactants). For example, consider the reactionN2(g)+3H2(g)â2NH3(g) for which În=2â(1+3)=â2. | Part A For the reaction2A(g)+2B(g)âC(g) Kc = 84.2 at a temperature of 163 âC . Calculate the value of Kp.Express your answer numerically. View Available Hint(s)
SubmitPrevious Answers Incorrect; Try Again; 5 attempts remaining Part B For the reactionX(g)+3Y(g)â3Z(g) Kp = 1.11Ã10â2 at a temperature of 153 âC . Calculate the value of Kc.Express your answer numerically. |