1
answer
0
watching
98
views
17 Nov 2019
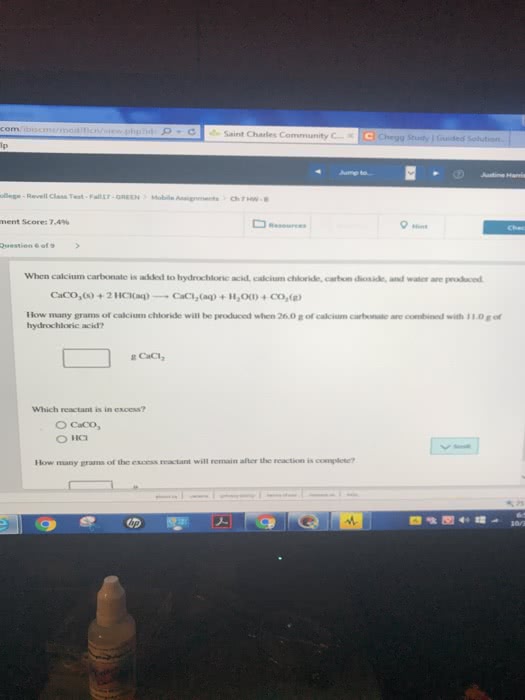
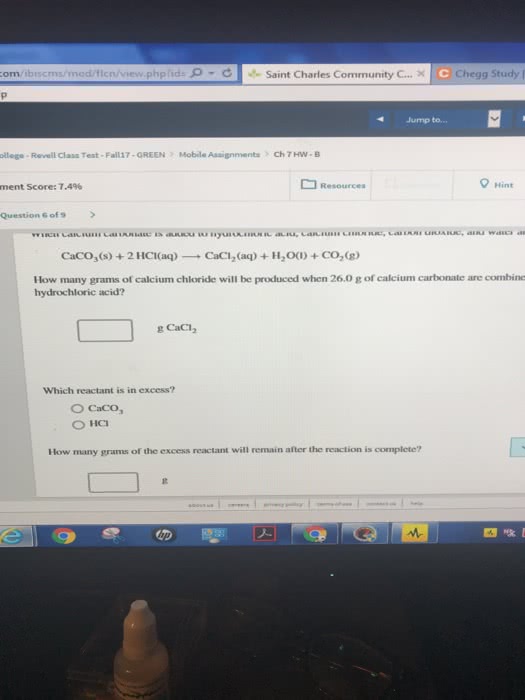
When calcium carbonate is added to hydrochloric acid, calcium chloride, carbon dioxide, and water are produced.
CaCO3 + 2HCl 
A) How many grams of calcium chloride will be produced when 26.0g of calcium carbonate are combined whith 13.0g of hydrochloric acid?
B) Which reactant is in excess and how many grams of this reactant will remain after the reaction is complete?
When calcium carbonate is added to hydrochloric acid, calcium chloride, carbon dioxide, and water are produced.
CaCO3 + 2HCl 
A) How many grams of calcium chloride will be produced when 26.0g of calcium carbonate are combined whith 13.0g of hydrochloric acid?
B) Which reactant is in excess and how many grams of this reactant will remain after the reaction is complete?
Keith LeannonLv2
10 Oct 2019