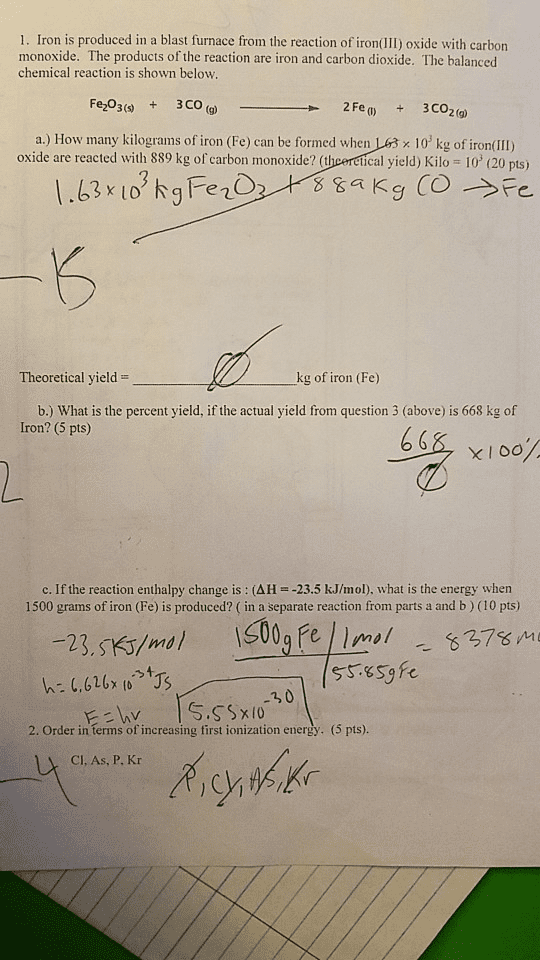1
answer
0
watching
206
views
17 Nov 2019


Iron is produced from by the reaction between carbon (coke) and iron (III) oxide: 2 Fe_2O_3 (s) + 3 C(s) rightarrow 4 Fe(s) + 3 CO_2(g) Upon reacting 530.0 kg of iron(III) oxide with a large excess of carbon, the amount of iron produced is determined to be 320.3 kg. The percent yield of iron is: 30.1% 35.0% 41.3% 43.2% 60.4% 62.9% 70.0% 86.4% None of the above; the correct yield is %
1
answer
0
watching
206
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Keith LeannonLv2
14 Feb 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232