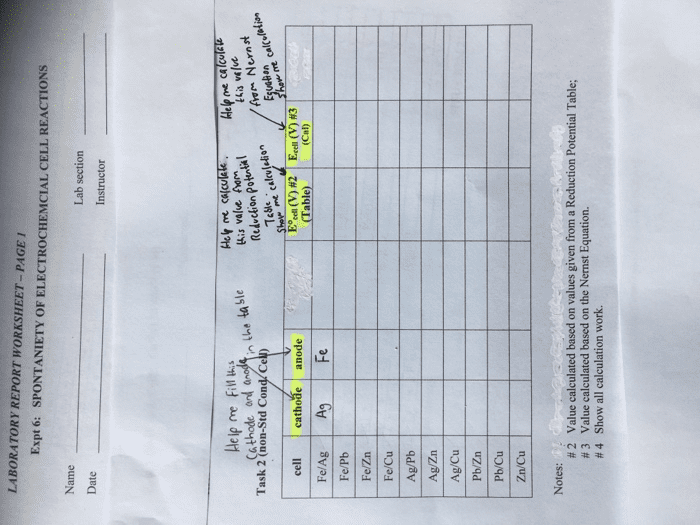
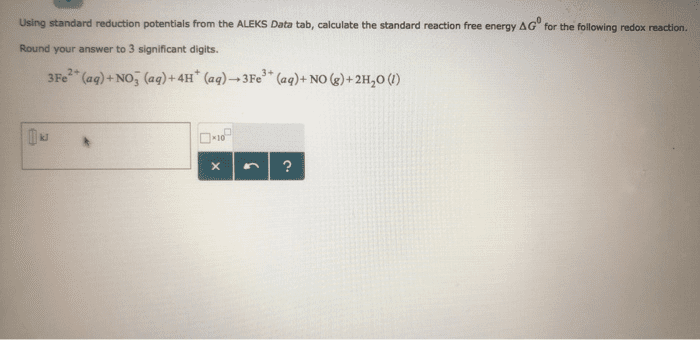
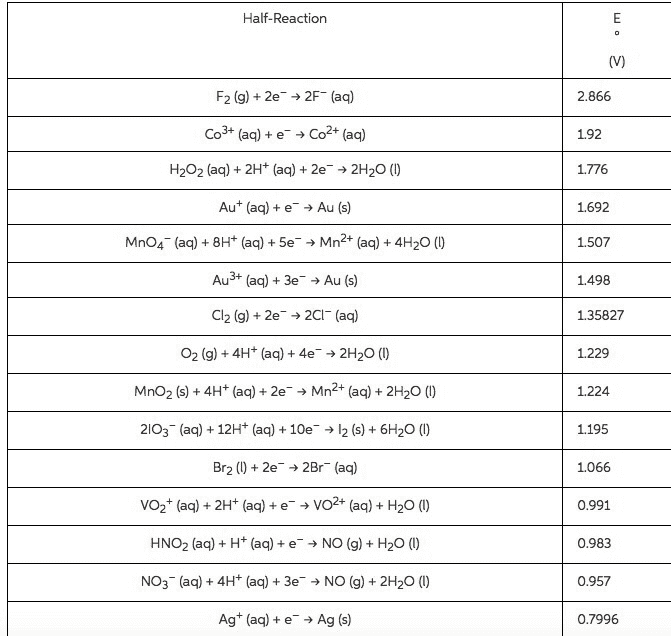
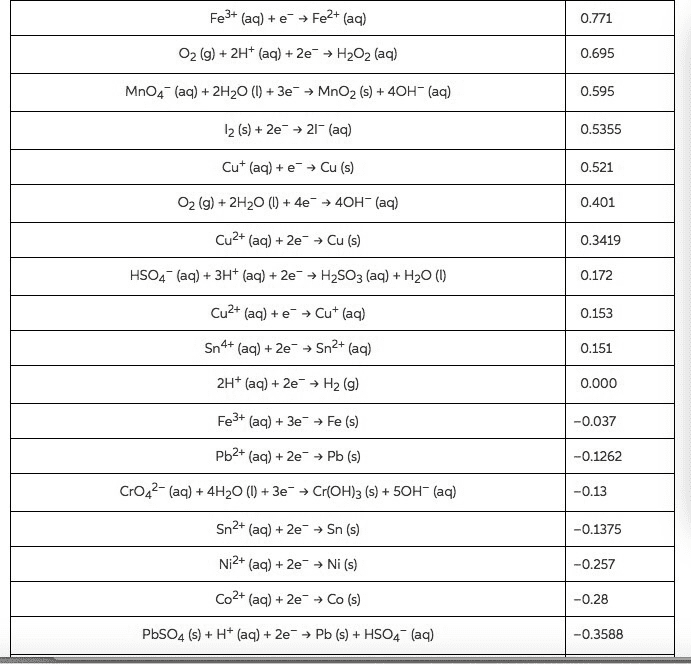
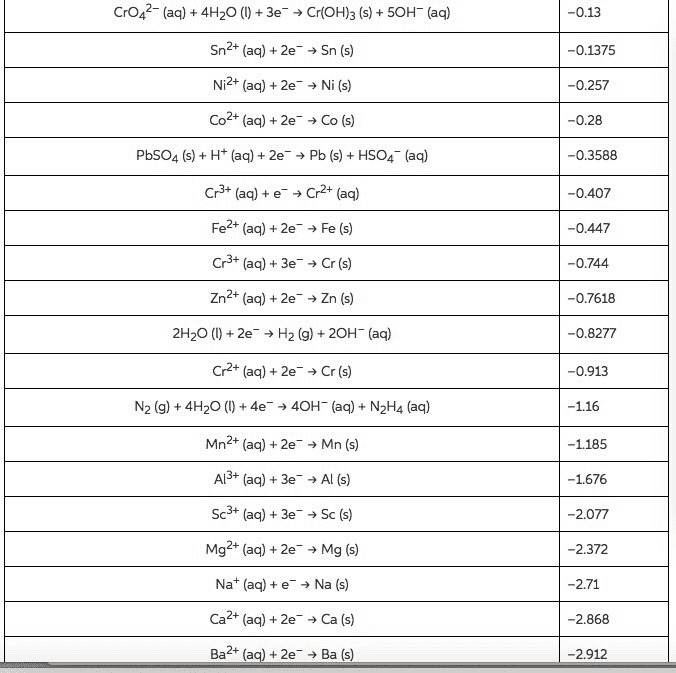
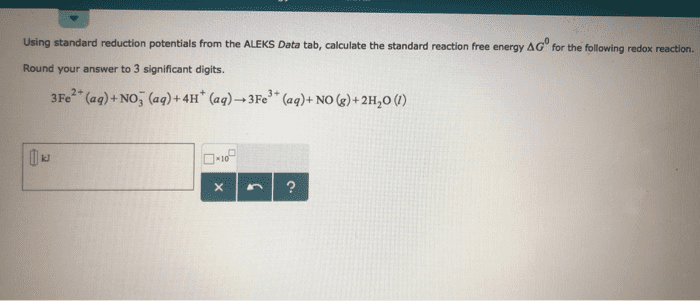
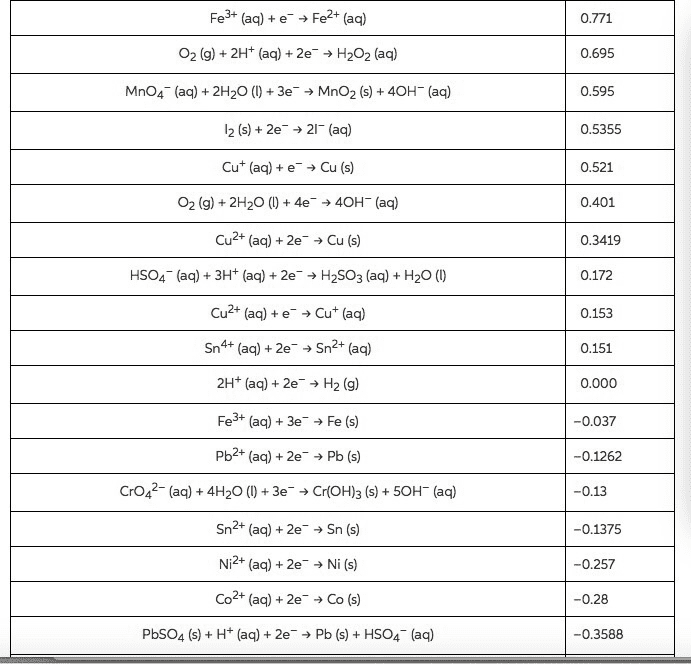
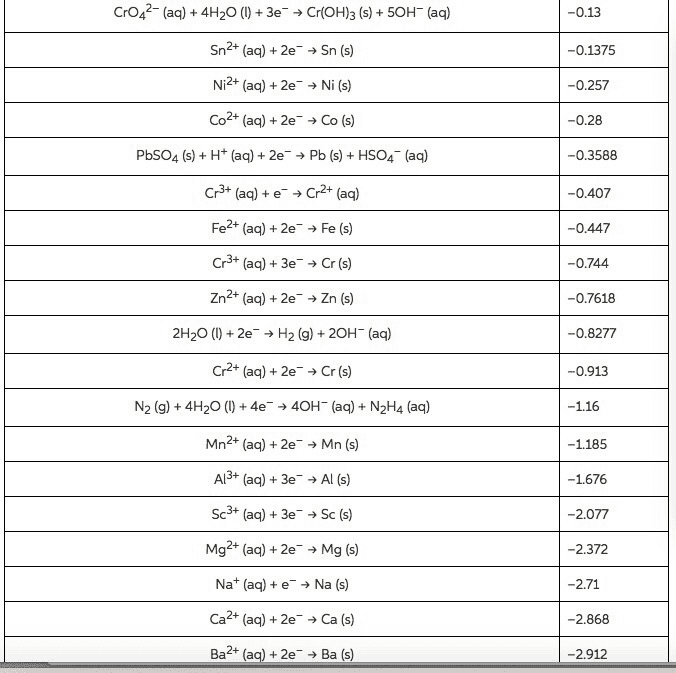
Use the Æ° from the standard reduction table to calculate âG° and Keq for each of the following reactions at 25°C (298K)
a) Pb+2(aq) + Mg(s)âPb(s) + Mg+2(aq)
b) Br2(l) + 2 Cl-(aq)âCl2(g) + 2 Br-(aq)
c) O2(g)+2 H2O(l) +2 Cu(s)â4 OH- (aq) + 2 Cu+2
d) MnO2 (s) + 4 H+(aq) +Cu(s)âMn+2(aq) + 2 H2O(l)+Cu+2(aq)
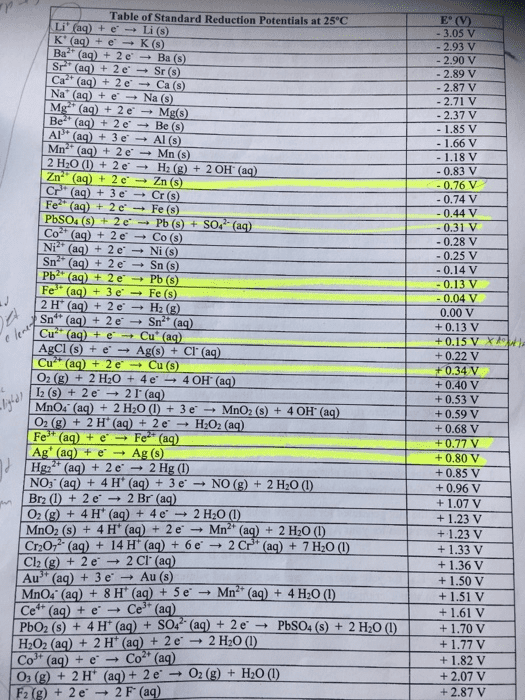
From standard reduction table:
Pb2+(aq) + 2e- â Pb(s) ε^ο(v) = -0.13
Mg2+(aq) + 2e- â Mg(s) ε^ο(v) = -2.37
Br2(l) + 2e- â 2Br-(aq) ε^ο(v) = +1.07
Cl2(g) + 2e- â 2Cl-(aq) ε^ο(v) = +1.36
O2(g) + 2H2O(l ) + 4e- â 4OH-(aq) ε^ο(v) = +0.40
Cu+2(aq) + 2e- â Cu(s) ε^ο(v) = +0.34
MnO2(s) + 4H+(aq) + 2e- â Mn+2(aq) + 2H2O(l) ε^ο(v) = +1.23
Cu+2(aq) + 2e- â Cu(s) ε^ο(v) = +0.34
Use the Æ° from the standard reduction table to calculate âG° and Keq for each of the following reactions at 25°C (298K)
a) Pb+2(aq) + Mg(s)âPb(s) + Mg+2(aq)
b) Br2(l) + 2 Cl-(aq)âCl2(g) + 2 Br-(aq)
c) O2(g)+2 H2O(l) +2 Cu(s)â4 OH- (aq) + 2 Cu+2
d) MnO2 (s) + 4 H+(aq) +Cu(s)âMn+2(aq) + 2 H2O(l)+Cu+2(aq)
From standard reduction table:
Pb2+(aq) + 2e- â Pb(s) ε^ο(v) = -0.13
Mg2+(aq) + 2e- â Mg(s) ε^ο(v) = -2.37
Br2(l) + 2e- â 2Br-(aq) ε^ο(v) = +1.07
Cl2(g) + 2e- â 2Cl-(aq) ε^ο(v) = +1.36
O2(g) + 2H2O(l ) + 4e- â 4OH-(aq) ε^ο(v) = +0.40
Cu+2(aq) + 2e- â Cu(s) ε^ο(v) = +0.34
MnO2(s) + 4H+(aq) + 2e- â Mn+2(aq) + 2H2O(l) ε^ο(v) = +1.23
Cu+2(aq) + 2e- â Cu(s) ε^ο(v) = +0.34
For unlimited access to Homework Help, a Homework+ subscription is required.