1
answer
0
watching
188
views
18 Nov 2019
Question 2
Part A
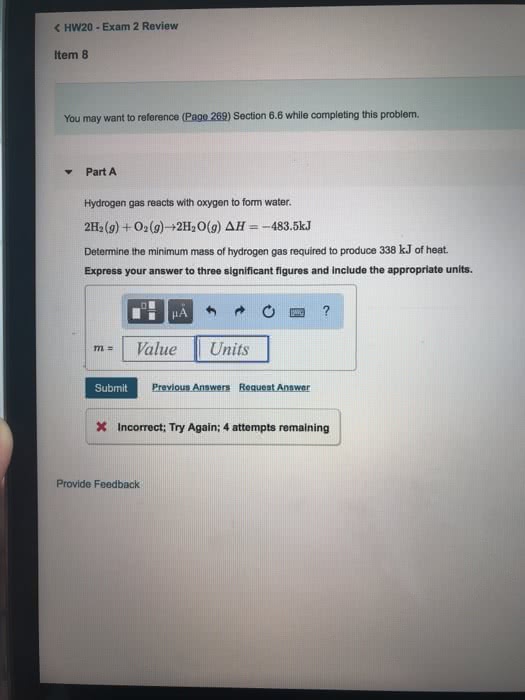
Hydrogen gas reacts with oxygen to form water.
2 H2(g) + O2(g) â 2 H2O(g) ÎH=â483.5kJ
What is the minimum mass of hydrogen gas required to produce 579 kJ of heat?
Express your answer to three significant figures and include the appropriate units.
Question 2
Part A
Hydrogen gas reacts with oxygen to form water.
2 H2(g) + O2(g) â 2 H2O(g) ÎH=â483.5kJ
What is the minimum mass of hydrogen gas required to produce 579 kJ of heat?
Express your answer to three significant figures and include the appropriate units.
Collen VonLv2
21 Apr 2019