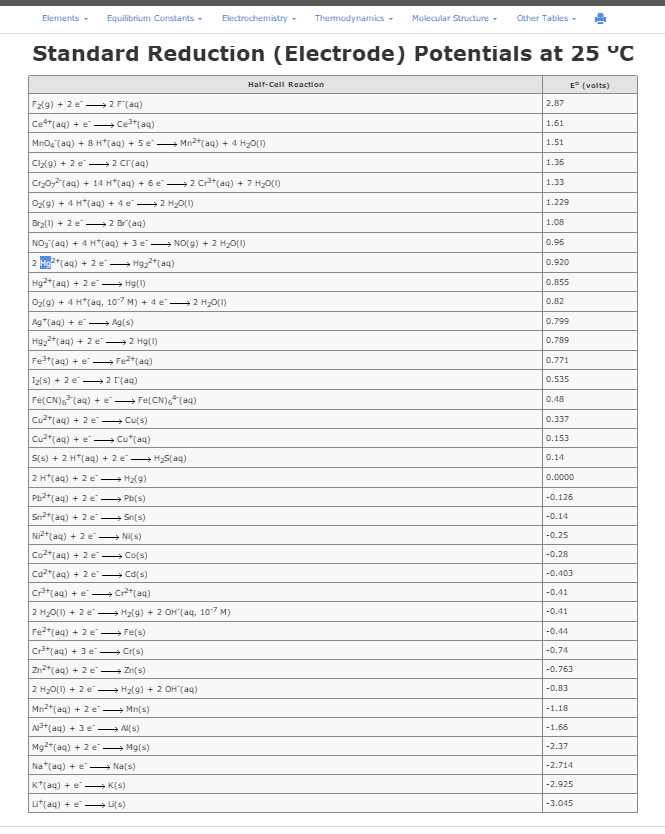
Which electrochemical reaction is spontaneous in a galvanic cell?
Ni2+(aq) + Cu(s) --> Ni(s) + Cu2+(aq)
Cr3+(aq) + Au(s) --> Cr(s) + Au3+(aq)
Cu2+(aq) + Co2+(aq) --> Cu+(aq) + Co3+(aq)
Cu2+(aq) + Sn(s) --> Cu(s) + Sn2+(aq)
Which electrochemical reaction is non-spontaneous in an electrolytic cell?
F2(g) + 2Cl-(aq) --> 2F-(aq) + Cl2(g)
Ag+(aq) + Fe2+(aq) --> Ag(s) + Fe3+(aq)
Co2+(aq) + Pb(s) --> Co(s) + Pb2+(aq)
Mn2+(aq) + Ba(s) --> Mn(s) + Ba2+(aq)
Which electrochemical reaction is the most spontaneous, generating the highest cell voltage?
F2(g) + 2Cl-(aq) --> 2F-(aq) + Cl2(g)
Ag+(aq) + Fe2+(aq) --> Ag(s) + Fe3+(aq)
Mn2+(aq) + Ba(s) --> Mn(s) + Ba2+(aq)
Cu2+(aq) + Sn(s) --> Cu(s) + Sn2+(aq)
Given the following notation for a galvanic cell diagram
Cd(s) â Cd2+(aq) â Pb2+(aq) â Pb(s)
Which one correctly represents the electrochemical reaction taking place in the galvanic cell?
Cd(s) + Pb2+(aq) --> Cd2+(aq) + Pb(s)
Cd2+(aq)+ Pb2+(aq) --> Cd(s)+ Pb(s)
Cd2+(aq) + Pb(s) --> Cd(s) + Pb2+(aq)
Cd2+(aq) + Pb2+(aq) --> Cd(s) + Pb2+(s)
Consider a Galvanic cell constructed from the following half cells, linked by a KCl salt bridge.
. an Al(s) electrode in 0.5 M Al2(SO4)3 solution
. a Pb(s) electrode in 1.0 M Pb(NO3)2 solution
Which electrode is the cathode?
Al
Pb
Al3+
Pb2+
Which electrochemical reaction is spontaneous in a galvanic cell?
| Ni2+(aq) + Cu(s) --> Ni(s) + Cu2+(aq) | ||
| Cr3+(aq) + Au(s) --> Cr(s) + Au3+(aq) | ||
| Cu2+(aq) + Co2+(aq) --> Cu+(aq) + Co3+(aq) | ||
| Cu2+(aq) + Sn(s) --> Cu(s) + Sn2+(aq) |
Which electrochemical reaction is non-spontaneous in an electrolytic cell?
| F2(g) + 2Cl-(aq) --> 2F-(aq) + Cl2(g) | ||||||||||||||||||||||||||||||||||||||
| Ag+(aq) + Fe2+(aq) --> Ag(s) + Fe3+(aq) | ||||||||||||||||||||||||||||||||||||||
| Co2+(aq) + Pb(s) --> Co(s) + Pb2+(aq) | ||||||||||||||||||||||||||||||||||||||
| Mn2+(aq) + Ba(s) --> Mn(s) + Ba2+(aq) Which electrochemical reaction is the most spontaneous, generating the highest cell voltage?
Given the following notation for a galvanic cell diagram Cd(s) â Cd2+(aq) â Pb2+(aq) â Pb(s) Which one correctly represents the electrochemical reaction taking place in the galvanic cell?
|