1
answer
0
watching
180
views
18 Nov 2019
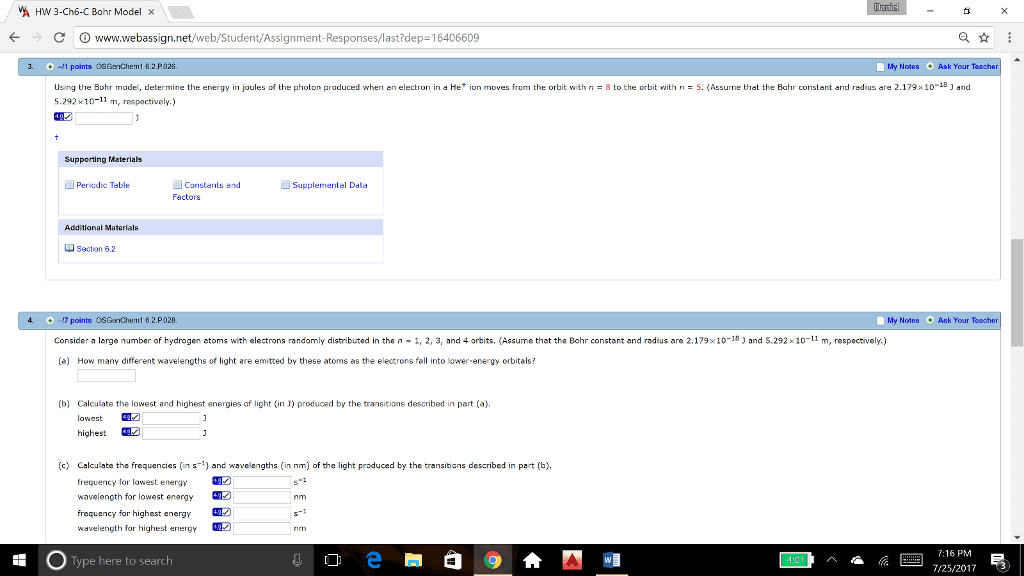
v A HW 3-Cho-C Bohr Model à â ã© â www.webassign.net/web/Student/Assignment.Responses/last?dep-16406609 My Notes Ask Your on moves from the orbit with n = 8 to the orbit with n = 5 Assume that the Bohr constant and radius are 2.179x10 18 J and Us ng the Bohr mode, determine the energy in joules of the photon produced when en electron in d He 5.292Ã10-11 m, respectively.) Supporting Materials Pericdic Table Constants and Factors Additional Materials section 6.2 4. 7 points OSGatChemt 8.2.P028. Consider a large number of hydrogen atoms with electrons randomly distributed in the n-1 2, 3, and 4 orbits. Assume that the Bohr constant and radius are 2.179Ã10-18 J and S 292 (a) How many different wavelengths of light are emitted by these atoms as the electrons fall into lower-energy orbitals? My Notes Ask Your 10-11 m, respect vely. (b) Calculate the lowest and highest energies of light (in J) produoed by the transitions described in part (a) lowest highest (c) Calculate the frequencies (in s1) and wavelengths (in nm) of the light produced by the transitians described in pert (b) y for lowest energy wavelength for lowest energy6 frequency for highest energy wavelength for highest energy2 7:16 PM R 25/20173 Type here to search

v A HW 3-Cho-C Bohr Model à â ã© â www.webassign.net/web/Student/Assignment.Responses/last?dep-16406609 My Notes Ask Your on moves from the orbit with n = 8 to the orbit with n = 5 Assume that the Bohr constant and radius are 2.179x10 18 J and Us ng the Bohr mode, determine the energy in joules of the photon produced when en electron in d He 5.292Ã10-11 m, respectively.) Supporting Materials Pericdic Table Constants and Factors Additional Materials section 6.2 4. 7 points OSGatChemt 8.2.P028. Consider a large number of hydrogen atoms with electrons randomly distributed in the n-1 2, 3, and 4 orbits. Assume that the Bohr constant and radius are 2.179Ã10-18 J and S 292 (a) How many different wavelengths of light are emitted by these atoms as the electrons fall into lower-energy orbitals? My Notes Ask Your 10-11 m, respect vely. (b) Calculate the lowest and highest energies of light (in J) produoed by the transitions described in part (a) lowest highest (c) Calculate the frequencies (in s1) and wavelengths (in nm) of the light produced by the transitians described in pert (b) y for lowest energy wavelength for lowest energy6 frequency for highest energy wavelength for highest energy2 7:16 PM R 25/20173 Type here to search
1
answer
0
watching
180
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
4 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232


