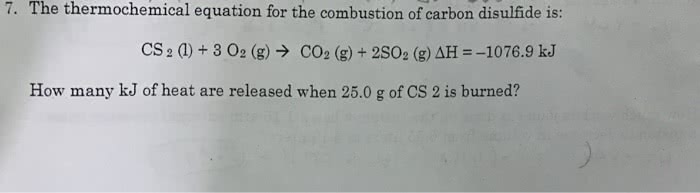1
answer
1
watching
910
views
18 Nov 2019
The combustion of one mole of liquid carbon disulfide, CS2 in oxygen produces carbon dioxide and sulfur dioxide gases and liberates 1075 kJ of heat.
A. write the thermo chemical equation for the combustion of the cabon disulfide.
B. what is delta H when 5.00 grams of CS2 must be burned to liberate 10.0 kJ of heat?
C. How many grams of CS2 must be burned to liberate 10.0 kJ of heat?
The combustion of one mole of liquid carbon disulfide, CS2 in oxygen produces carbon dioxide and sulfur dioxide gases and liberates 1075 kJ of heat.
A. write the thermo chemical equation for the combustion of the cabon disulfide.
B. what is delta H when 5.00 grams of CS2 must be burned to liberate 10.0 kJ of heat?
C. How many grams of CS2 must be burned to liberate 10.0 kJ of heat?
Deanna HettingerLv2
20 May 2019