1
answer
0
watching
97
views
18 Nov 2019

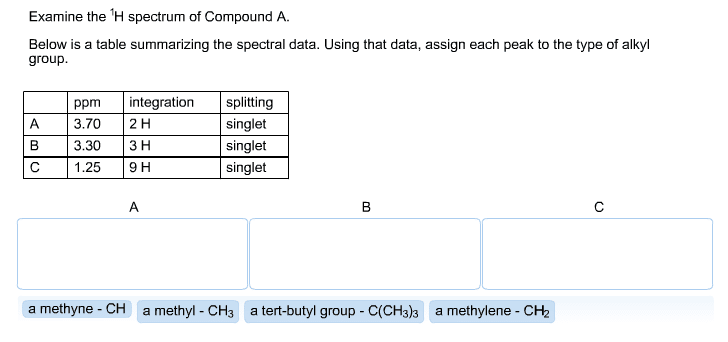
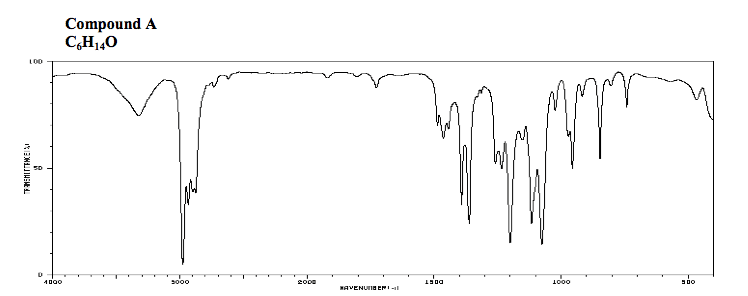
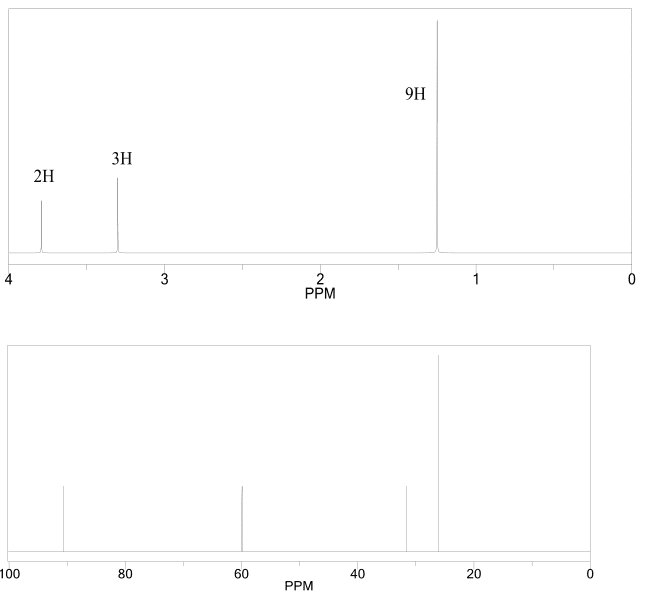
What would be the expected reaction outcome if you ran the reaction in concentrated H_2 SO_4; keeping in mind that HSO_4^- is a poor nucleophile? The JR and molecular formula of compound is provided below. Calculate degree of unsaturation of the compound and characterized the compound by IR.

What would be the expected reaction outcome if you ran the reaction in concentrated H_2 SO_4; keeping in mind that HSO_4^- is a poor nucleophile? The JR and molecular formula of compound is provided below. Calculate degree of unsaturation of the compound and characterized the compound by IR.
1
answer
0
watching
97
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Beverley SmithLv2
15 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232