1
answer
0
watching
428
views
18 Nov 2019
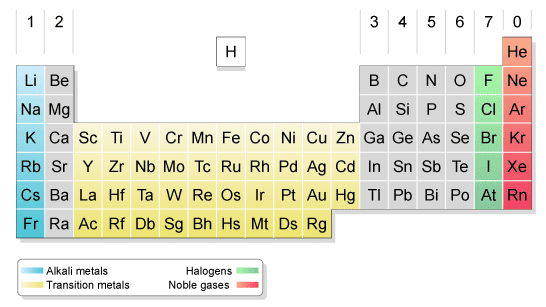
(4) For each of the elements Ca, K, Cu, Zn, Br, and Kr, provide the following information: (a) Which are metals? (b) Which are main-group metals? (c) Which are inert or noble gases?
(5) Using the periodic table, write the electron configuration and orbital diagram of each of the following atoms: (a) B; (b) S; (c) Ar.
(6) Which of the following pairs of atoms and / or ions are isoelectronic with one another?
(a) Fâ, Clâ (b) K+, Ar
(7) Arrange each of the following lists of elements in order of increasing atomic size:
(a) N, O, F (b) Li, K, Cs (c) Cl, Br, I
(4) For each of the elements Ca, K, Cu, Zn, Br, and Kr, provide the following information: (a) Which are metals? (b) Which are main-group metals? (c) Which are inert or noble gases?
(5) Using the periodic table, write the electron configuration and orbital diagram of each of the following atoms: (a) B; (b) S; (c) Ar.
(6) Which of the following pairs of atoms and / or ions are isoelectronic with one another?
(a) Fâ, Clâ (b) K+, Ar
(7) Arrange each of the following lists of elements in order of increasing atomic size:
(a) N, O, F (b) Li, K, Cs (c) Cl, Br, I
Lelia LubowitzLv2
8 Jun 2019