0
answers
0
watching
89
views
18 Nov 2019
--------------------SKIP NH3------------------------------------
SOLVE THE TWO QUESTIONS BELOW
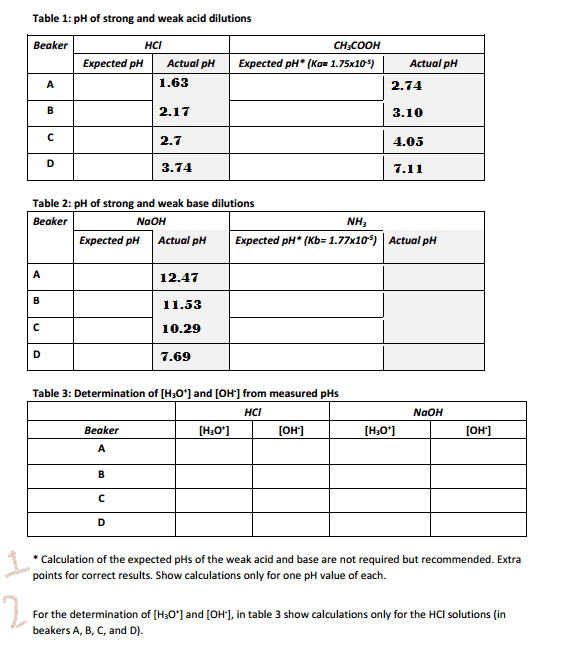
Calculation of the expected pHs of the weak acid and base are not required but recommended. Extra points for correct results. Show calculations only for one pH value of each. For the determination of [H_3O^+] and [OH^-], in table 3 show calculations only for the HCl solutions (in beakers A, B, C, and D).
--------------------SKIP NH3------------------------------------
SOLVE THE TWO QUESTIONS BELOW

Calculation of the expected pHs of the weak acid and base are not required but recommended. Extra points for correct results. Show calculations only for one pH value of each. For the determination of [H_3O^+] and [OH^-], in table 3 show calculations only for the HCl solutions (in beakers A, B, C, and D).
0
answers
0
watching
89
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232



