1
answer
0
watching
339
views
18 Nov 2019
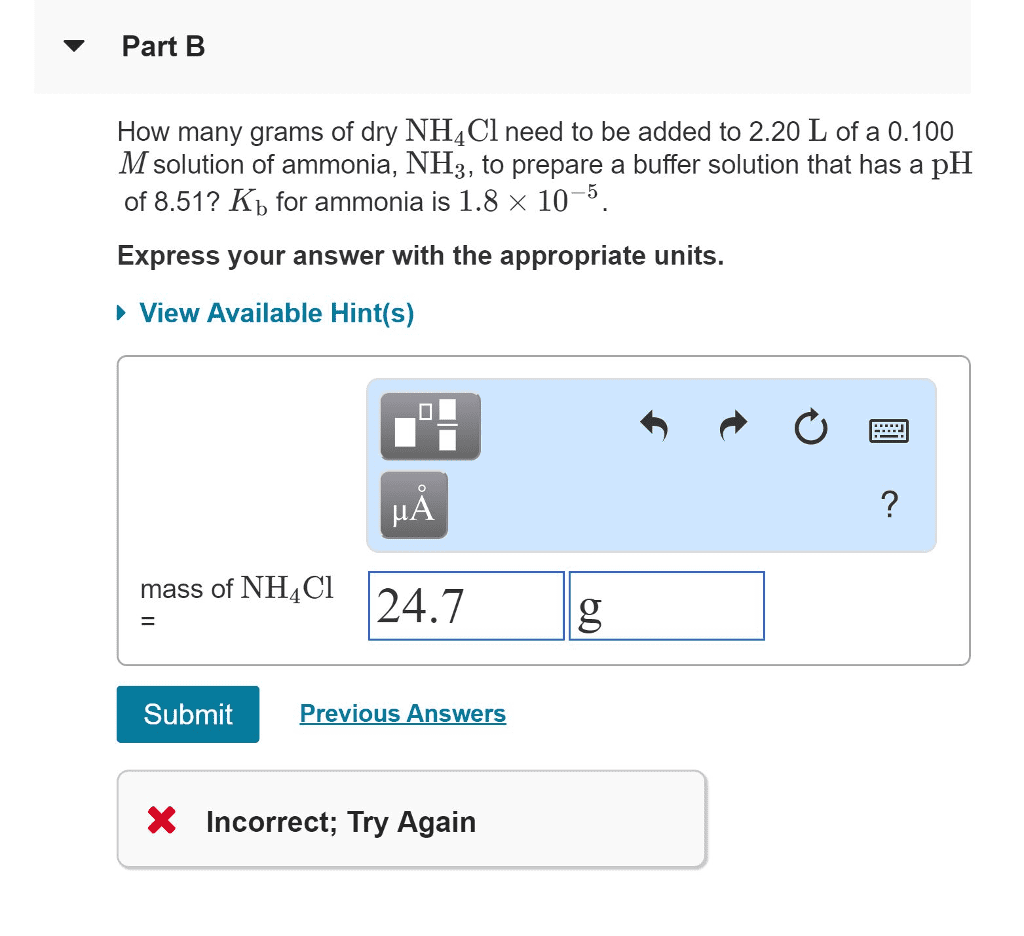
How many grams of dry NH4Cl need to be added to 2.40 L of a 0.100 M solution of ammonia, NH3, to prepare a buffer solution that has a pH of 8.52? Kb for ammonia is 1.8Ã10â5.
Just as pH is the negative logarithm of [H3O+], pKa is the negative logarithm of Ka,
pKa=âlogKa
The Henderson-Hasselbalch equation is used to calculate the pH of buffer solutions:
pH=pKa+log[base]/[acid]
Notice that the pH of a buffer has a value close to the pKaof the acid, differing only by the logarithm of the concentration ratio [base]/[acid]. The Henderson-Hasselbalch equation in terms of pOH and pKb is similar.
pOH=pKb+log[acid]/[base]
best answer will be rated. Thank you for your time in advance!!
How many grams of dry NH4Cl need to be added to 2.40 L of a 0.100 M solution of ammonia, NH3, to prepare a buffer solution that has a pH of 8.52? Kb for ammonia is 1.8Ã10â5.
Just as pH is the negative logarithm of [H3O+], pKa is the negative logarithm of Ka,
pKa=âlogKa
The Henderson-Hasselbalch equation is used to calculate the pH of buffer solutions:
pH=pKa+log[base]/[acid]
Notice that the pH of a buffer has a value close to the pKaof the acid, differing only by the logarithm of the concentration ratio [base]/[acid]. The Henderson-Hasselbalch equation in terms of pOH and pKb is similar.
pOH=pKb+log[acid]/[base]
best answer will be rated. Thank you for your time in advance!!
Jarrod RobelLv2
10 Sep 2019