1
answer
0
watching
2,339
views
18 Nov 2019
Classify each chemical reaction as a combination, precipitation, single replacement, combustion, double replacement, acid-base, decomposition.
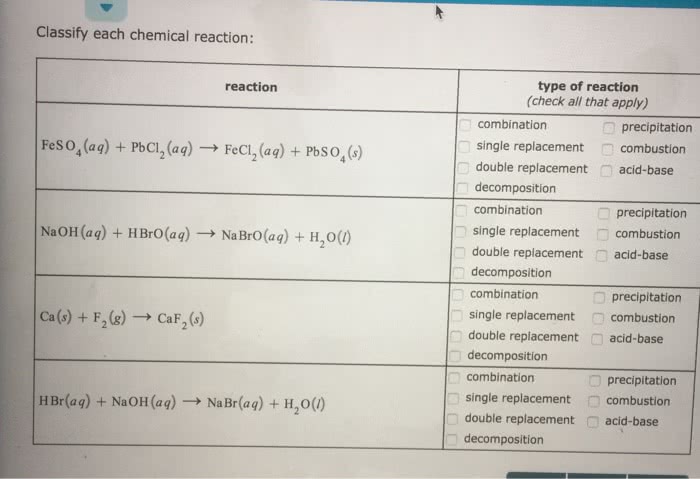
Classify each chemical reaction as a combination, precipitation, single replacement, combustion, double replacement, acid-base, decomposition.


Classify each chemical reaction: type of reaction (check all that apply) reaction combination single replacement combustion double replacement acid-base precipitation Feso,(aq) + PbCl2(aq) â FeCl2(aq) + PbSO4(s) -1 decomposition combination precipitation -) single replacement © combustion NaOH (aq) + H Bro(aq) â NaBrO(aq) + H20(1) acid-base double replacement decomposition combination precipitation combustion -single replacement Ca (s) + F 2(g) â CaF2(s) double replacement acid-base ä¸decomposition combination precipitation single replacement combustion H Br(aq) + NaOH (aq) â Na Br(aq) + H2O(1) -double replacement acid-base decomposition
Hubert KochLv2
8 Feb 2019


