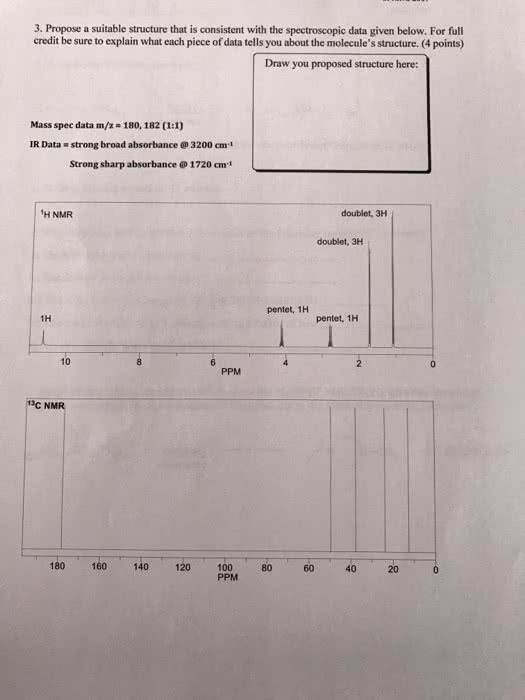
Be sure to answer all parts.
Consider the following SN2 reaction:

What happens to the reaction rate in each of the following instances?
Part 1:
The leaving group is changed from Brâ to Iâ.
The reaction is
not changedfasterslower
.
Part 2:
The solvent is changed from acetone to
CH3CH2OH
.
The reaction rate
increasesdecreasesstays the same
.
Part 3:
The alkyl halide is changed from
CH3(CH2)4Br
to
CH3CH2CH2CH(Br)CH3.
The reaction rate
increasesdecreasesstays the same
.
Part 4:
The concentration of âCN is increased by a factor of 9.
Reaction rate will
decreaseincrease
by a factor of

.
Part 5:
The concentration of both the alkyl halide and âCN are increased by a factor of 3.
Reaction rate will
increase 
by a factor of ............?????? I got this one wrong. What is it? ( THIS IS THE ONLY ONE I GOT WRONG IN All THE PARTS)
Br+ CN CN + Br acetone
Be sure to answer all parts.
Consider the following SN2 reaction:

What happens to the reaction rate in each of the following instances?
| Part 1: |
The leaving group is changed from Brâ to Iâ.
The reaction is
not changedfasterslower
.
| Part 2: |
The solvent is changed from acetone to
CH3CH2OH
.
The reaction rate
increasesdecreasesstays the same
.
| Part 3: |
The alkyl halide is changed from
CH3(CH2)4Br
to
CH3CH2CH2CH(Br)CH3.
The reaction rate
increasesdecreasesstays the same
.
| Part 4: |
The concentration of âCN is increased by a factor of 9.
Reaction rate will
decreaseincrease
by a factor of

.
| Part 5: |
The concentration of both the alkyl halide and âCN are increased by a factor of 3.
Reaction rate will
increase 
by a factor of ............?????? I got this one wrong. What is it? ( THIS IS THE ONLY ONE I GOT WRONG IN All THE PARTS)