For the reaction shown, calculate how many grams of oxygen form when each quantity of reactant completely reacts. 2HgO(s)â2Hg(l)+O2(g) Part A 2.67 gHgO m = g SubmitMy AnswersGive Up Part B 6.84 gHgO m = g SubmitMy AnswersGive Up Part C 1.78 kgHgO m = g SubmitMy AnswersGive Up Part D 3.28 mgHgO
For the reaction shown, calculate how many grams of oxygen form when each quantity of reactant completely reacts. 2HgO(s)â2Hg(l)+O2(g) Part A 2.67 gHgO m = g SubmitMy AnswersGive Up Part B 6.84 gHgO m = g SubmitMy AnswersGive Up Part C 1.78 kgHgO m = g SubmitMy AnswersGive Up Part D 3.28 mgHgO
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
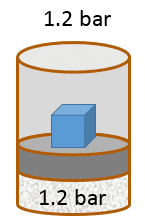
| pV work - Exercise 2 This problem continues Exercise 1. However, note that the initial volume and temperature may have changed due to variable randomization. Pay attention to the values stated below and use them in all your calculations for this excercise. In exercise 1 the piston was allowed to move freely. Now, you put stoppers on the piston so it cannot move up or down.
Remember to use three significant figures for all numerical answers. The margin of error for each numerical answer is 5%. To avoid rounding errors use the unrounded intermediate values in your final calculations. The initial volume of gas is 4.20 L , the initial temperature of the gas is 32.0 °C , and the system is in equilibrium with an external pressure of 1.2 bar (given by the sum of a 1 bar atmospheric pressure and a 0.2 bar pressure due to a brick that rests on top of the piston). Then, as you did in Exercise 1, you heat the gas slowly until the temperature reaches 57.2 °C | Part A How many moles of gas are there in the container?
SubmitMy AnswersGive Up Part B What is the final volume after you reach the final temperature?
SubmitMy AnswersGive Up Part C What is the final pressure after you reach the final temperature?
SubmitMy AnswersGive Up Part D What is the sign of w? What is the sign of w?
SubmitMy AnswersGive Up Part E What is the value of w?
SubmitMy AnswersGive Up |
1. For the reaction, calculate how many moles of the product form when 1.77mol of Na completely reacts. Assume that there is more than enough of the other reactant. 2Na(s)+O2(g)--->Na2O2(s)
2. For the reaction, calculate how many moles of the product form when 2.69mol of O2 completely reacts.
Assume that there is more than enough of the other reactant.
2S(s)+3O2(g)?2SO3(g)
| 3. For the reaction shown, calculate how many grams of oxygen form when each quantity of reactant completely reacts. 2HgO(s)?2Hg(l)+O2(g) a) 2.58 gHgO b) 6.03 gHgO c) 1.92 kgHgO d) 3.67 mgHgO |
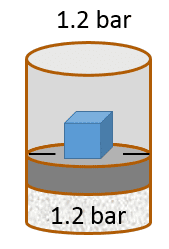
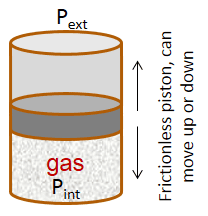
| pV work - Exercise 1 Consider a container with a frictionless piston that contains a given amount of an ideal gas. If the external pressure is kept constant, the piston will move up or down in response to a change in the internal pressure. The piston will move up if Pint > Pext and vice versa. The piston will stop moving when Pint = Pext (the system is equilibrated).
The following problem will slowly guide you through a work calculation. Some questions may appear trivial, so trust your instincts and donât assume there are hidden tricks. The goal of such a guided exercise is to help you picture the situation so you understand how all relevant variables change or stay the same. This is the first in a sequence of related problems. Remember to use three significant figures for all numerical answers. The margin of error for each numerical answer is 5%. To avoid rounding errors use the unrounded intermediate values in your final calculations. Letâs assume the initial volume of the gas is 3.90 L , the initial temperature of the gas is 26.0 °C , and the system is in equilibrium with an external pressure of 1.2 bar (given by the sum of a 1 bar atmospheric pressure and a 0.2 bar pressure due to a brick that rests on top of the piston).
| Part A How many moles of gas are there in the container?
SubmitMy AnswersGive Up You now heat the gas slowly until the temperature reaches 51.2 °C You do it slowly so the piston can move if necessary until a new equilibrium is reached. Part B What happens with the piston while you heat the gas? What happens with the piston while you heat the gas?
When the final temperature is reached, you stop heating the gas. Part C What is the final pressure of the gas?
SubmitMy AnswersGive Up Part D What is the final volume of the gas?
SubmitMy AnswersGive Up Part E What happens with the piston after you finish heating the gas? Assume you do not need to worry about the gas cooling down again because the outside of the container is at a lower temperature. That is, you manage to keep the gas at a constant temperature that equals 51.2 °C What happens with the piston after you finish heating the gas? Assume you do not need to worry about the gas cooling down again because the outside of the container is at a lower temperature. That is, you manage to keep the gas at a constant temperature that equals 51.2
SubmitMy AnswersGive Up So far this was all at the level of your general chemistry class. Letâs now calculate the work involved in the process. Recall the sign conventions we use in physical chemistry, and always think from the point of view of the system (the gas). Part F What is the sign of w? What is the sign of w?
SubmitMy AnswersGive Up Part G What is the value of w? Be careful with units. How do you convert bar*L to J?
SubmitMy AnswersGive Up |