1
answer
0
watching
264
views
18 Nov 2019
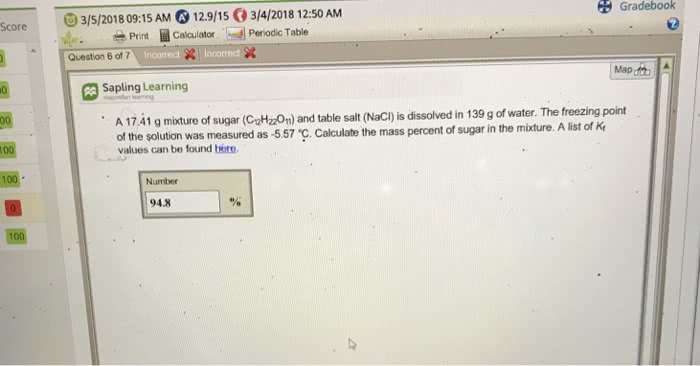
A 16.06 g mixture of sugar (C12H22O11) and table salt (NaCl) is dissolved in 195 g of water. The freezing point of the solution was measured as -3.75 °C. Calculate the mass percent of sugar in the mixture. A list of Kf values can be found here.
A 16.06 g mixture of sugar (C12H22O11) and table salt (NaCl) is dissolved in 195 g of water. The freezing point of the solution was measured as -3.75 °C. Calculate the mass percent of sugar in the mixture. A list of Kf values can be found here.
Trinidad TremblayLv2
13 Oct 2019