1
answer
0
watching
523
views
18 Nov 2019
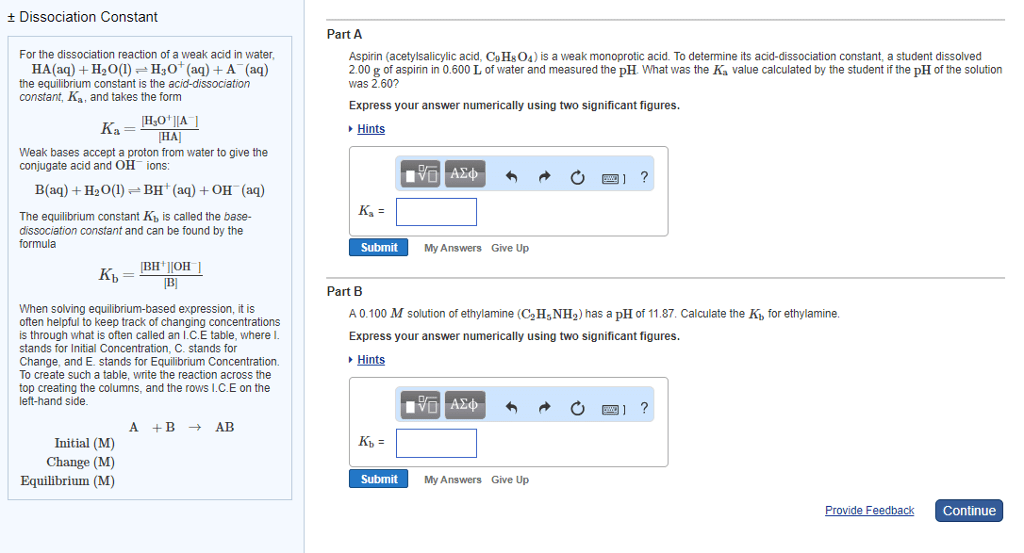
Part A Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 L of water and measured the pH. What was the Ka value calculated by the student if the pH of the solution was 2.60? Express your answer numerically using two significant figures. Hints Ka = SubmitMy AnswersGive Up Part B A 0.100 M solution of ethylamine (C2H5NH2) has a pH of 11.87. Calculate the Kb for ethylamine. Express your answer numerically using two significant figures. Hints Kb = SubmitMy AnswersGive Up
Part A Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 L of water and measured the pH. What was the Ka value calculated by the student if the pH of the solution was 2.60? Express your answer numerically using two significant figures. Hints Ka = SubmitMy AnswersGive Up Part B A 0.100 M solution of ethylamine (C2H5NH2) has a pH of 11.87. Calculate the Kb for ethylamine. Express your answer numerically using two significant figures. Hints Kb = SubmitMy AnswersGive Up
Elin HesselLv2
8 Jul 2019