1
answer
0
watching
189
views
18 Nov 2019
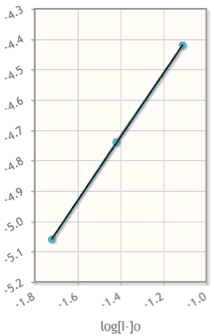
Hi, so I am having a hard time creating the rate law because I can not figure out how to find the reaction order. I keep ending up with .5 = 5^x. Please help! The test is tomorrow
Experiment Initial Concentration CO (M) Initial Concentration NO2 (M) Initial Rate of Reaction (M/sec) 1 5.0x10^-4 3.6x10^-5 3.4x10^-8 2 5.0x10^-4 1.8x10^-5 1.7x10^-8 3 1.0x10^-3 3.6x10^-5 6.8x10^-8 4 1.5x10^-3 7.2x10^-5 ?
A. What is the rate law for this reaction? Make sure to include the units for k in your work.
B. What is the initial rate of the reaction in Experiment 4?
Hi, so I am having a hard time creating the rate law because I can not figure out how to find the reaction order. I keep ending up with .5 = 5^x. Please help! The test is tomorrow
| Experiment | Initial Concentration CO (M) | Initial Concentration NO2 (M) | Initial Rate of Reaction (M/sec) |
|---|---|---|---|
| 1 | 5.0x10^-4 | 3.6x10^-5 | 3.4x10^-8 |
| 2 | 5.0x10^-4 | 1.8x10^-5 | 1.7x10^-8 |
| 3 | 1.0x10^-3 | 3.6x10^-5 | 6.8x10^-8 |
| 4 | 1.5x10^-3 | 7.2x10^-5 | ? |
A. What is the rate law for this reaction? Make sure to include the units for k in your work.
B. What is the initial rate of the reaction in Experiment 4?
1
answer
0
watching
189
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Casey DurganLv2
31 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232