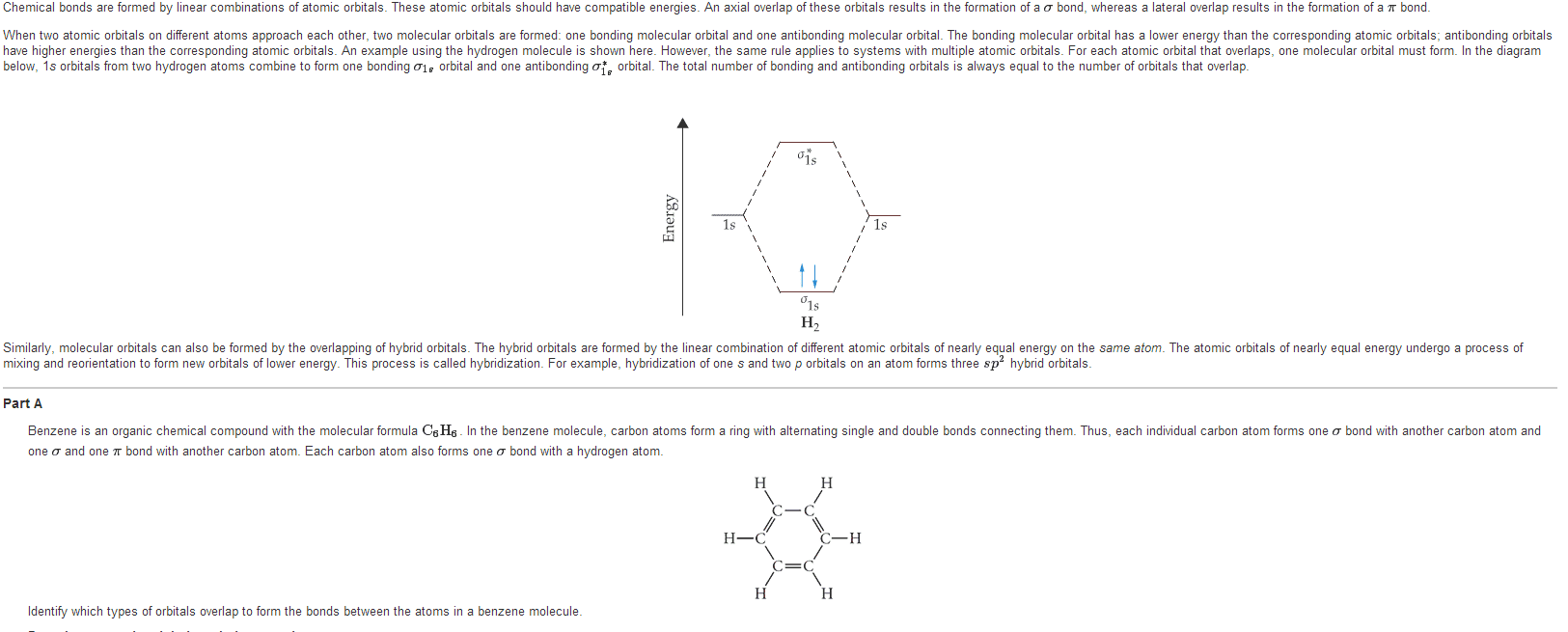
1
answer
0
watching
311
views
19 Nov 2019

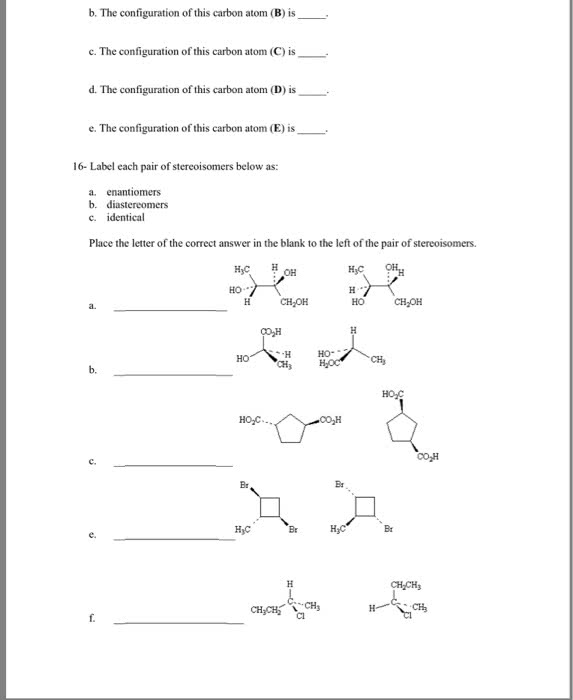
3. For each of the following molecules, circle all chiral carbons. Also, indicate whether the molecule will rotate plane polarized light. Remember that each intersection of lines is a carbon atom and each carbon atom will have a total of four bonds. When 4 bonds are not shown, the remaining bonds to carbon are to an appropriate number of hydrogen atoms. CH3 OH CH NH2 ilj NH2 b) OH c)

3. For each of the following molecules, circle all chiral carbons. Also, indicate whether the molecule will rotate plane polarized light. Remember that each intersection of lines is a carbon atom and each carbon atom will have a total of four bonds. When 4 bonds are not shown, the remaining bonds to carbon are to an appropriate number of hydrogen atoms. CH3 OH CH NH2 ilj NH2 b) OH c)
1
answer
0
watching
311
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Reid WolffLv2
31 May 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232