1
answer
0
watching
112
views
19 Nov 2019
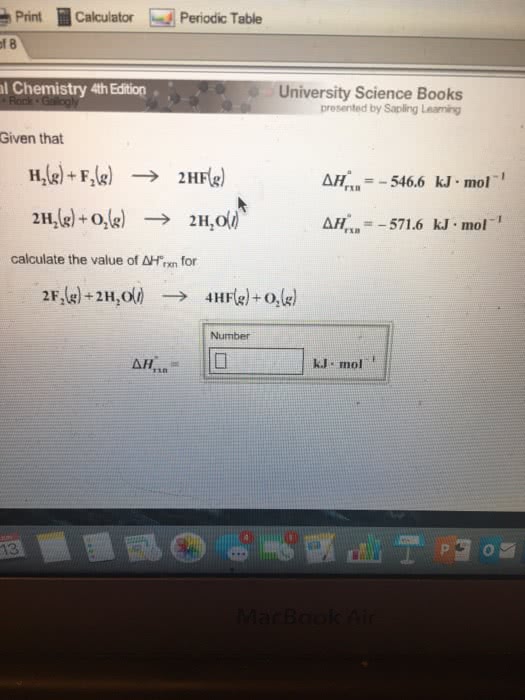

Given that H_2(g) + F_2(g) rightarrow 2HF(g) Delta H degree_rxn = 546.6 kJ middot mol^-1 2H_2(g) + O_2(g) rightarrow 2H_2O(l) Delta degree _rxn = -571.6 kJ middot mol^-1 calculate the value of Delta H degree_rxn for 2F_2(g) + 2H_2O(l) rightarrow 4HF(g) + O_2(g) Delta H degree _rxn = kJ middot mol^-1
Lelia LubowitzLv2
11 May 2019
