1
answer
0
watching
1,054
views
19 Nov 2019
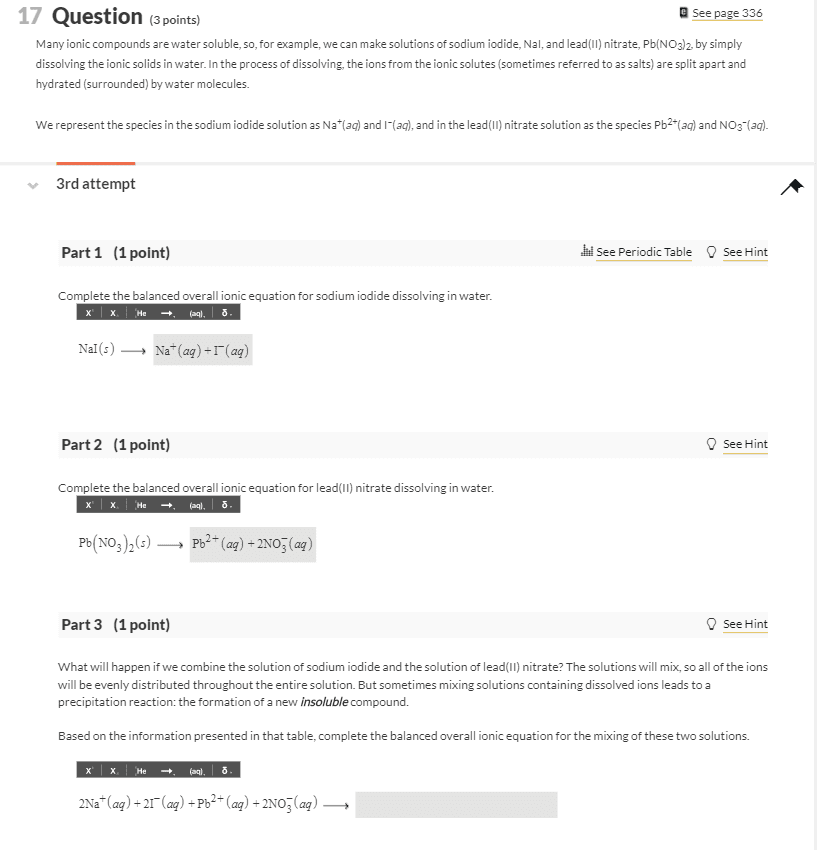
When ionic solids dissolve in water, they produce soluble aqueous cations and anions. For example, adding MgBr2 solids to water will produce ions according to the dissolution reaction below. MgBr2(s) â Mg2+(aq) + 2 Brâ(aq) solid cation anion In this lab, you will be mixing two solutions containing soluble ions and observing any reactions that occur. Please write the dissolution reaction for each of the ionic solids below. (Use the lowest possible coefficients. Include states-of-matter under the given conditions in your answer.)
(a) Na3PO4
(b) Mg(NO3)2
(c) Pb(NO3)2
When ionic solids dissolve in water, they produce soluble aqueous cations and anions. For example, adding MgBr2 solids to water will produce ions according to the dissolution reaction below. MgBr2(s) â Mg2+(aq) + 2 Brâ(aq) solid cation anion In this lab, you will be mixing two solutions containing soluble ions and observing any reactions that occur. Please write the dissolution reaction for each of the ionic solids below. (Use the lowest possible coefficients. Include states-of-matter under the given conditions in your answer.)
(a) Na3PO4
(b) Mg(NO3)2
(c) Pb(NO3)2
1
answer
0
watching
1,054
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
3 Jul 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232