1
answer
1
watching
352
views
19 Nov 2019
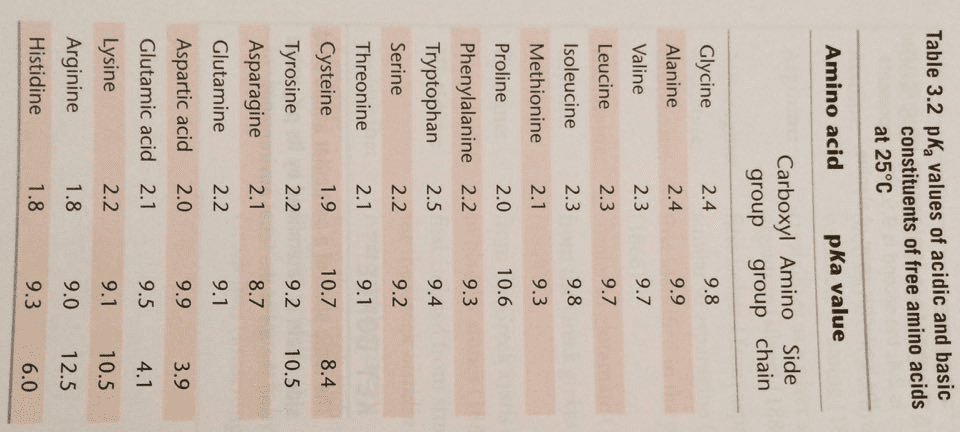
What is the isoelectric point (pI) of the following peptide? To facilitate answering this question, assume that the N-terminal aminium group has a pKa of 8, the C-terminal carboxyl group has a pKa of 3.5, and that the ionizable side chains have pKas as follows: Asp = 4.0, Glu = 4.0, His = 6, Cys = 8.4, Tyr = 10, Lys = 10, and Arg = 12.5. Also, assume that if the pH is at least two units above or below the pKa of a particular group, then that group is completely unprotonated or protonated, as the case may be.
What is the isoelectric point (pI) of the following peptide? To facilitate answering this question, assume that the N-terminal aminium group has a pKa of 8, the C-terminal carboxyl group has a pKa of 3.5, and that the ionizable side chains have pKas as follows: Asp = 4.0, Glu = 4.0, His = 6, Cys = 8.4, Tyr = 10, Lys = 10, and Arg = 12.5. Also, assume that if the pH is at least two units above or below the pKa of a particular group, then that group is completely unprotonated or protonated, as the case may be.
Elin HesselLv2
27 Feb 2019