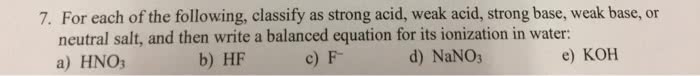1
answer
1
watching
1,027
views
19 Nov 2019
Which of the following statements concerning aqueous solutions of salts is FALSE?
A) For any salt of a strong acid and a strong base, the pH will be about 7.
B) For any salt of a strong acid and a weak base, the pH will be <7.
C) For any salt of a weak acid and a strong base, the pH will be >7.
D) For any salt of a weak acid and a weak base, the pH will be about 7.
SA/SB=Neutral, SA/WB=Acidic, WA/SB=?, WA/WB=? Please help :)
Which of the following statements concerning aqueous solutions of salts is FALSE?
A) For any salt of a strong acid and a strong base, the pH will be about 7.
B) For any salt of a strong acid and a weak base, the pH will be <7.
C) For any salt of a weak acid and a strong base, the pH will be >7.
D) For any salt of a weak acid and a weak base, the pH will be about 7.
SA/SB=Neutral, SA/WB=Acidic, WA/SB=?, WA/WB=? Please help :)
Jarrod RobelLv2
6 Feb 2019