1
answer
0
watching
845
views
20 Nov 2019
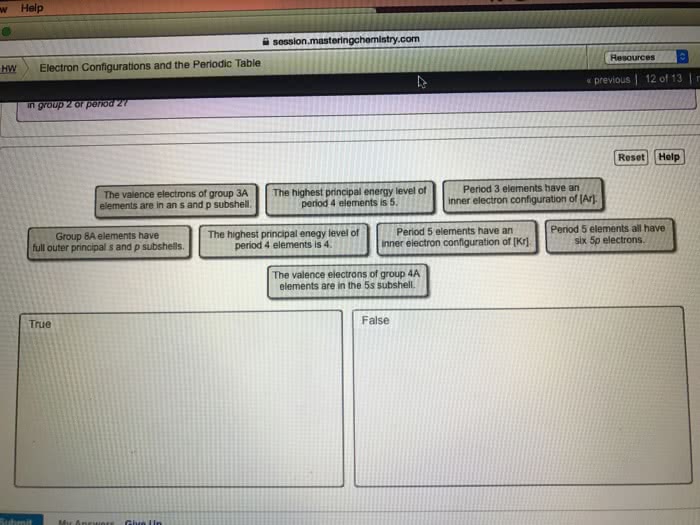
Classify each statement as true or false.1. Period 4 elements have an inner electron configuration of Ar.2. Group 8A elements have full outer principle s and p subshells.3 Period 5 elements have an inner electron configuration of Xe.4. The highest principle quantum number of period 3 elements is 4.5. Period 3 elements have six 2p electrons.6. The valence electrons of group 2A elements are in an s subshell.7. The valence electrons of group 5A elemnts are in the 6s subshell.8. The highest principle quantum number of period 2 elements is 2.
Classify each statement as true or false.1. Period 4 elements have an inner electron configuration of Ar.2. Group 8A elements have full outer principle s and p subshells.3 Period 5 elements have an inner electron configuration of Xe.4. The highest principle quantum number of period 3 elements is 4.5. Period 3 elements have six 2p electrons.6. The valence electrons of group 2A elements are in an s subshell.7. The valence electrons of group 5A elemnts are in the 6s subshell.8. The highest principle quantum number of period 2 elements is 2.
Patrina SchowalterLv2
19 Jun 2019