1
answer
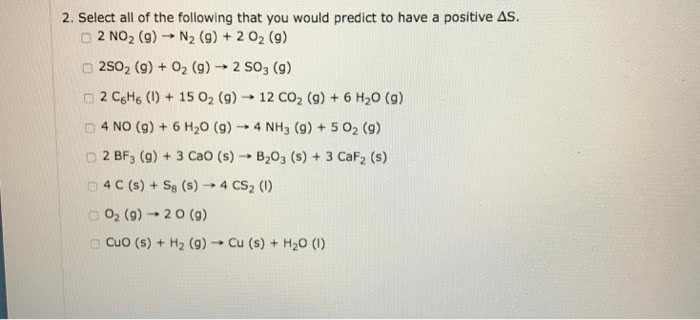
0
watching
236
views
23 Nov 2019
Consider the reaction:
8 H2S(g) + 4 O2(g) ----> 8 H2O(g) + S8(g)
let delta = d, molarity = M, seconds = s
d[H2S]/dt = -0.080 M/s
Find:
a) d[O2]/dt
b) d[H2O]/dt
c) d[S8]/dt
d) Rate
Please show all work and show complete steps to find thesolution.
Consider the reaction:
8 H2S(g) + 4 O2(g) ----> 8 H2O(g) + S8(g)
let delta = d, molarity = M, seconds = s
d[H2S]/dt = -0.080 M/s
Find:
a) d[O2]/dt
b) d[H2O]/dt
c) d[S8]/dt
d) Rate
Please show all work and show complete steps to find thesolution.
Trinidad TremblayLv2
9 Aug 2019