0
answers
0
watching
129
views
23 Nov 2019
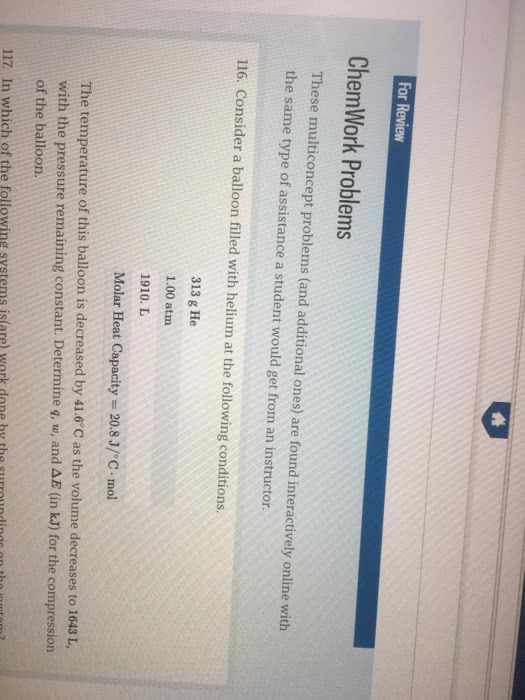
A balloon filled with 34.3mol helium has a volume of 768.L at 0.0°C and 1.00 atm pressure. The temperature of the balloon isincreased to 33.0°C as it expands to a volume of 861. L, the pressure remaining constant.Calculate q, w, andÎE forthe helium in the balloon. (The molar heat capacity for helium gasis 20.8 J/°C · mol.)
q ________ kJ w ________kJ ÎE ________ kJ
A balloon filled with 34.3mol helium has a volume of 768.L at 0.0°C and 1.00 atm pressure. The temperature of the balloon isincreased to 33.0°C as it expands to a volume of 861. L, the pressure remaining constant.Calculate q, w, andÎE forthe helium in the balloon. (The molar heat capacity for helium gasis 20.8 J/°C · mol.)
| q | ________ kJ |
| w | ________kJ |
| ÎE | ________ kJ |