1
answer
0
watching
122
views
ochreclam516Lv1
29 Sep 2019
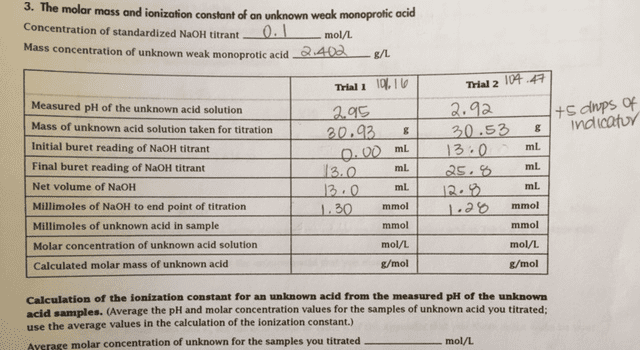
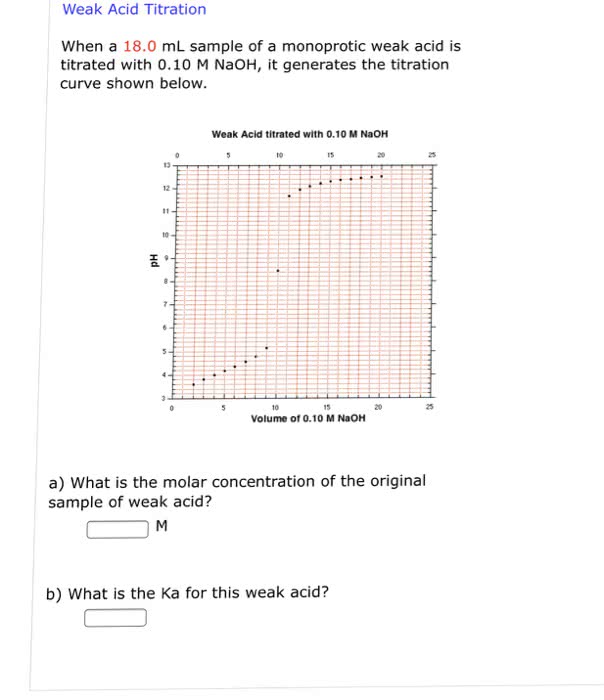
A weak monoprotic acid is titrated with NaOH, and the followingdata are recorded. Calculate the molar mass of the weak acid.
original volume of acid solution = 50.00 mL mass of acid (a solid)= 0.3459 g
Volume of NaOH titrant required: 34.72 mL concentration of NaOH =0.09951 M
A weak monoprotic acid is titrated with NaOH, and the followingdata are recorded. Calculate the molar mass of the weak acid.
original volume of acid solution = 50.00 mL mass of acid (a solid)= 0.3459 g
Volume of NaOH titrant required: 34.72 mL concentration of NaOH =0.09951 M
original volume of acid solution = 50.00 mL mass of acid (a solid)= 0.3459 g
Volume of NaOH titrant required: 34.72 mL concentration of NaOH =0.09951 M
Hubert KochLv2
29 Sep 2019