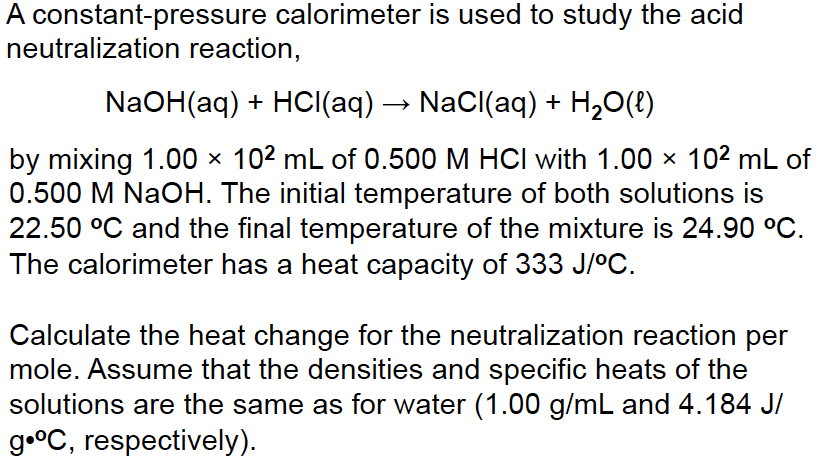In a coffee cup calorimeter, 20.0 mL of 2.00 M NaOH and 20.0 mL of2.00 M HCl are mixed. Both solutions were originally at 22.4 C.After the reaction, the final temperature is 33.8 C.
HCl (aq) + NaOH(aq) H2O(l) + NaCl(aq) + heat.
Heat lost by the reaction + heat gained by the NaCl solution =0
q lost + q gained = 0 q gained = (s) (m) (?T)
(A) Calculate the volume of NaCl(aq) (this will be the volume ofthe solution in the calorimeter).
(B) Given that the density of 1.00 M NaCl is 1.03 g/mL, calculatethe mass of the NaCl(aq).
(C) Calculate the ?T for the NaCl(aq).
(D) Given that the specific heat capacity of 1.00 M NaCl(aq) is3.87 J/gC, calculate the heat gained by the solution (qgained)
q gained
Given that 25.0 mL of 2.00 M NaOH and 25.0 mL of 2.00 M HClreacted. Calculate the number of moles of either NaOH or HCl thatreacted. Hneutralization is the heat for the reaction in units ofkJ/mole.
Calculate Hneutralization. (Pay close attention to the sign)
Hneutralization
In a coffee cup calorimeter, 20.0 mL of 2.00 M NaOH and 20.0 mL of2.00 M HCl are mixed. Both solutions were originally at 22.4 C.After the reaction, the final temperature is 33.8 C.
HCl (aq) + NaOH(aq) H2O(l) + NaCl(aq) + heat.
Heat lost by the reaction + heat gained by the NaCl solution =0
q lost + q gained = 0 q gained = (s) (m) (?T)
(A) Calculate the volume of NaCl(aq) (this will be the volume ofthe solution in the calorimeter).
(B) Given that the density of 1.00 M NaCl is 1.03 g/mL, calculatethe mass of the NaCl(aq).
(C) Calculate the ?T for the NaCl(aq).
(D) Given that the specific heat capacity of 1.00 M NaCl(aq) is3.87 J/gC, calculate the heat gained by the solution (qgained)
q gained
Given that 25.0 mL of 2.00 M NaOH and 25.0 mL of 2.00 M HClreacted. Calculate the number of moles of either NaOH or HCl thatreacted. Hneutralization is the heat for the reaction in units ofkJ/mole.
Calculate Hneutralization. (Pay close attention to the sign)
Hneutralization