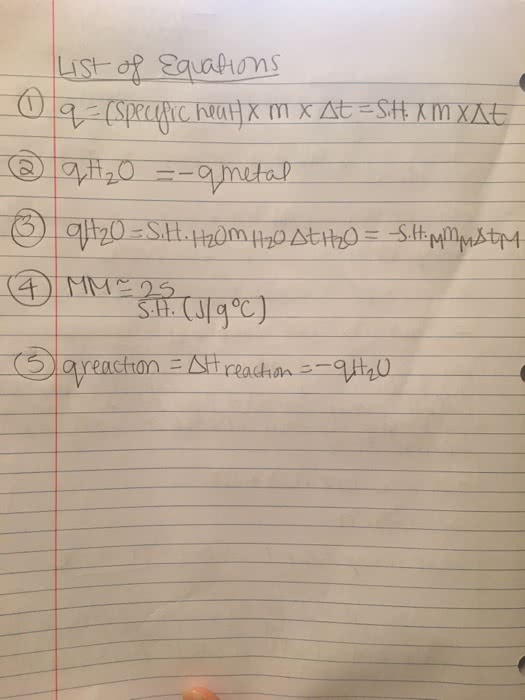1
answer
0
watching
317
views
29 Sep 2019
A metal sample weighing 45.0g and at a temperature of 99.1degrees Cwas placed in 38.6g of water in a calorimeter at 25.0 degrees C.The calorimeter reached a maximum temperature of 32.8 degreesC.
a. What was the change in temp?
b. What was the change in temp. of the metal?
c. How much heat flowed into the water?
d. Calculate the specific heat capacity of the metal? c=4.18
e. What is the approximate molar mass of the metal?
A metal sample weighing 45.0g and at a temperature of 99.1degrees Cwas placed in 38.6g of water in a calorimeter at 25.0 degrees C.The calorimeter reached a maximum temperature of 32.8 degreesC.
a. What was the change in temp?
b. What was the change in temp. of the metal?
c. How much heat flowed into the water?
d. Calculate the specific heat capacity of the metal? c=4.18
e. What is the approximate molar mass of the metal?
a. What was the change in temp?
b. What was the change in temp. of the metal?
c. How much heat flowed into the water?
d. Calculate the specific heat capacity of the metal? c=4.18
e. What is the approximate molar mass of the metal?
Liked by 14.kayla.18
Irving HeathcoteLv2
29 Sep 2019