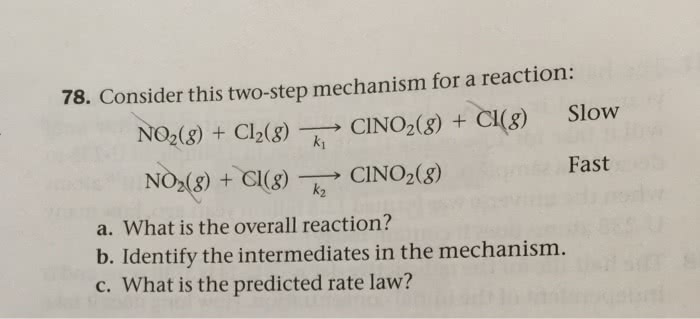Three part rate law question:) Will always thumbs up if the answer is right. Thanks again!
Part a)
Consider the overall reaction: AB(g) + CB(g) ⶠA(g) + CB2(g)
A proposed mechanism for this reaction is
2 AB(g) â AB2(g) + A(g) (fast)
AB2(g) + CB(g) ⶠAB(g) + CB2(g) (slow)
Based on this mechanism, determine the rate law for the overall reaction.
Part b)
Consider the following three-step mechanism for a reaction:
Br2 (g) â 2 Br (g) Fast
Br (g) + CHBr3 (g) â HBr (g) + CBr3 (g) Slow
Br (g) + CBr3 (g) â CBr4 (g) Fast
Based on this mechanism, determine the rate law for the overall reaction.
Part c)
Phosgene (Cl2CO), a poison gas used in World War I, is formed by the reaction of Cl2 and CO. The proposed mechanism for the reaction is
Cl2 â 2 Cl (fast,equilibrium)
Cl + CO â ClCO (fast,equilibrium)
ClCO + Cl2 â Cl2CO + Cl (slow)
Based on this mechanism, determine the rate law for the overall reaction.
Express your answers in terms of k and the necessary concentrations (ex: k[X][Y]^2). Use brackets around the concentrations and the caret symbol prior to the exponent. Do not include any multiplication signs.
Three part rate law question:) Will always thumbs up if the answer is right. Thanks again!
Part a)
Consider the overall reaction: AB(g) + CB(g) ⶠA(g) + CB2(g)
A proposed mechanism for this reaction is
2 AB(g) â AB2(g) + A(g) (fast)
AB2(g) + CB(g) ⶠAB(g) + CB2(g) (slow)
Based on this mechanism, determine the rate law for the overall reaction.
Part b)
Consider the following three-step mechanism for a reaction:
Br2 (g) â 2 Br (g) Fast
Br (g) + CHBr3 (g) â HBr (g) + CBr3 (g) Slow
Br (g) + CBr3 (g) â CBr4 (g) Fast
Based on this mechanism, determine the rate law for the overall reaction.
Part c)
Phosgene (Cl2CO), a poison gas used in World War I, is formed by the reaction of Cl2 and CO. The proposed mechanism for the reaction is
Cl2 â 2 Cl (fast,equilibrium)
Cl + CO â ClCO (fast,equilibrium)
ClCO + Cl2 â Cl2CO + Cl (slow)
Based on this mechanism, determine the rate law for the overall reaction.
Express your answers in terms of k and the necessary concentrations (ex: k[X][Y]^2). Use brackets around the concentrations and the caret symbol prior to the exponent. Do not include any multiplication signs.